http://www.chemistrymag.org/cji/2008/108038pe.htm
Aug.
1, 2008 Vol.10 No.8 P.38 Copyright ![]()
http://www.chemistrymag.org/cji/2008/108038pe.htm |
Aug.
1, 2008 Vol.10 No.8 P.38 Copyright |
1. INTRODUCTION
Congo Red is a low molecular weight anionic secondary diazo dye used in variety of
products such as consumable goods, textile and leather etc
2. EXPERIMENTAL
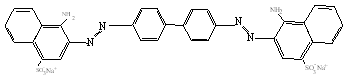
Congo Red (CR) (Mw=696.66),
purity > 99% and PAH (Mw =70,000), purity >99%, were purchased from
Aldrich Chemical Co. and were used as received. The chemical structures of PAH and CR are
shown in figure 1. The sequential adsorption of CR and PAH from dilute solution to form
PAH-CR multilayer was carried out by the same procedure presented in previous work [12].
The electrolytic deposition bath was prepared with 103 M aqueous solution using
triple distilled deionised (18.2 MW) Millipore water. A thoroughly cleaned quartz substrate
was first immersed into PAH aqueous solution for 15 minutes. After rinsing 3 times in
Milli Q water (2 minutes each bath with agitation), the modified substrate was then
transferred into the CR solution for 15 minutes, agitated and washed 3 times with water
for 2 minutes. All the adsorption procedures were carried out at room temperature
(approximately 250C). The repetition of the whole sequence produced desired
number of bi-layers of ELBL films. The characterizations of PAH-CR ELBL films have been
done by UV-Vis absorption (Lambda -25 UV-Vis spectrometer, Perkin Elmer)
 ˇˇ
ˇˇ 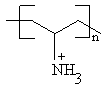
Fig.1 The chemical structures of PAH and CR
3. RESULTS AND DISCUSSION
Figure 2 shows the UV-Vis absorption spectra of aqueous solution of CR (1 X 10-5)
and 1 bilayer ELBL film of PAH-CR along with microcrystal (MC) of CR. The upper spectrum
is the second derivative of the spectrum of 1 bilayer of PAH-CR film.
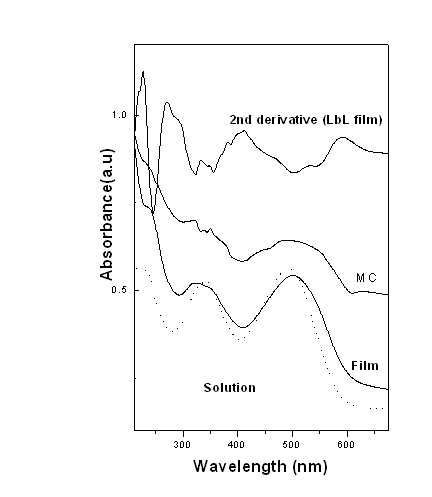
Fig. 2 The UV-Vis absorption spectra of aqueous solution of CR (1 X 10-5)
and 1 bilayer ELBL film of PAH-CR along with microcrystal (MC) of CR. The upper spectrum
is the second derivative of the spectrum of 1 bilayer of PAH-CR film.
From the calculation of
Edwards and Woods[14]it was
found that the band at 339 nm is due to transition moment parallel to the short axis of CR
and that for 500 nm band the transition moments are parallel to long axis of the molecule.
Since there is a strong appearance of band at 520 nm in the ELBL film, it suggests that
the CR molecules are aligned in such a way that the transition moments are along the
longer axis.It is observed that the solution spectra has a shorter wavelength at 340 nm
where as the band gets shifted to 350 nm in both ELBL films and MC. The maximum absorption
of CR is at 495 nm and there is a red shift of about 15 nm compared with that of PAH-CR
bilayer film. This spectroscopic red shift has been reported earlier and it is attributed
to the formation of well ordered J aggregates in the film domain [15]. The band at 510 nm is a longer wavelength band,
second derivative analysis revealed hidden component at 520 nm which is absent in the
solution spectra. This may be due to the fact that in solution of CR molecules are in
"monomer" form, minor band found at 520 nm in case of film can be related to CR
molecules are in "associated" form. This
appearance of new band is useful for finding thee molecular association of CR species.
According to the molecular excition theory [18] the energy difference between the excited
monomer level and the exciton level is given by
DE=M2 (1-3Cos q)/R3
Where M is the electric dipole transition moment and R is
the distance between the molecules. Here is the angle between transition dipole moment and
the molecular bonding axis. If 00<q<54.70 , the excition level is energetically located
below the monomer level causing red shift in the electronic absorption spectrum creating J
aggregates[19].So by molecular excition theory the red shift in
the spectra can be identified as formation of J aggregates .
The location of the 520 nm band being close to that of the band
exhibited by aggregated species can be said due to dominance of associated species over
the monomeric species in the ELBL films. A schematic representation of the interaction
scheme between PAH and CR molecules is shown in figure 3 .The interaction scheme reveals
that the anionic part of CR molecules interact with the cationic part of the PAH molecule
forming complex species. In the complex species CR and PAH molecules get closer side by
side and closer association of CR molecules take place which might cause the shift of
absorption band in the PAH-RB ELBL films.
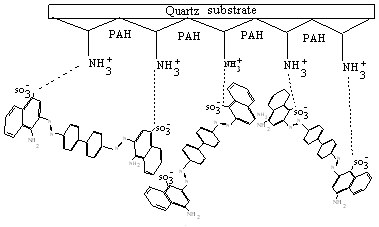
Fig.3 A schematic representation of the interaction scheme between PAH and CR molecules.
3.1 Layer effect
Figure 4 shows the UV-Vis absorption spectra for the multilayer prepared by sequential
deposition of PAH and CR from their dilute solution. It was observed that the absorption
spectra of different layered (1- bilayers) PAH-CR ELBL Self assembled films show almost
similar band pattern with characteristic peak at 350 nm and 510 nm irrespective of the
layer number except an increase in absorption intensity. The characteristic peaks at 350
nm and 510 nm is due to P-P*
transition[16]. Inset (a) of figure 4 is the plot of absorbance at
510 nm as a function of numbers of bilayer. A linear increase of intensity with a slope of
0.0051 O.D/ bilayer was observed which indicated that the sequential growth of PAH-CR
assemblies was linear and reproducible.
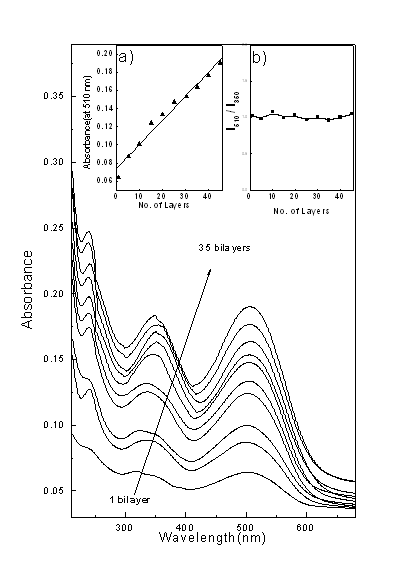
Fig. 4 shows the UV-Vis absorption spectra for the multilayer prepared by sequential deposition of PAH and CR from their dilute solution. Inset (a) is the plot of absorbance at 510 nm as a function of numbers of bilayer. Inset (b) the ratio of intensities of aggregate and monomeric band as a function of number of layers deposited.
The electrostatic interaction between PAH and sulfonate
anion of CR is crucial driving force in the formation of PAH-CR films. The formation of J
aggregates among CR molecules leads to a considerable increase in the dye content in the
composite films.
In order to know
whether the nature of aggregation has changed as deposition progressed, we have plotted
the ratio of intensities of aggregate and monomeric band as a function of number of layers
deposited (Inset b of Figure 4). The ratio remained almost same for all the number of
layers which indicated that there is no change of nature of aggregation during the
deposition process.
3.2 Binding and material loss
The binding property and material loss of PAH¨CCR
films prepared on the quartz surface was evaluated by UV-Vis absorption spectra of films
after dipping in CR solution and PAH solution.
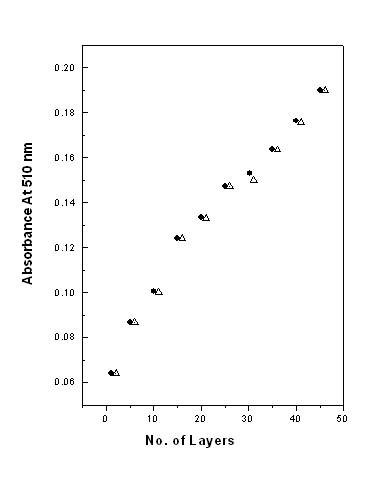
Fig. 5 shows the intensity of
absorption maximum at 510 nm as a function of number of deposited layers. The (ˇő) represent absorption maximum
recorded after deposition of CR and (D) represent the absorption maximum after each layer
deposition of polycation (PAH)
Figure 5
shows the intensity of absorption maximum at 510 nm as a function of number of deposited
layers. The (-ˇő-)
represent absorption maximum recorded after deposition of CR and (--D--) represent the
absorption maximum after each layer deposition of polycation (PAH). From the Figure it is
evident that the absorption maxima increases after each CR layer and remained almost same
after deposition of each PAH layer. This confirms that the CR molecules do not come out of
the film during PAH deposition, which implies that the binding between PAH and CR
molecules is very strong and thus PAH-CR ELBL films are fabricated almost without any
material loss.
3.3 Adsorption dynamics
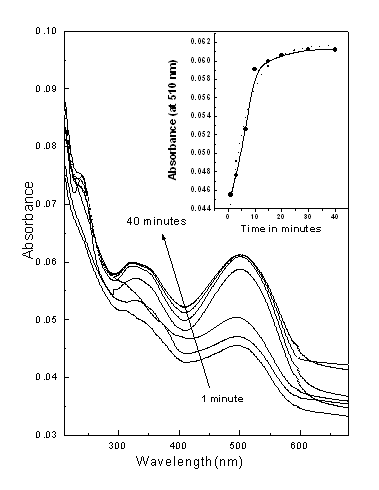
Fig.6 The adsorption of CR on the PAH films
at different immersion times. Inset of figure 6 is a plot of the maximum absorbance as a
function of time. The experimental result (solid line) and the dotted line in the inset of
figure 6 show the curve generated by fitting to the second order kinetic rate equation.
Figure 6 shows the adsorption of CR on the PAH films at different immersion
times. The adsorption takes place at relatively fast rate in first 10 minutes. The fast
rate of adsorption initially is probably due to the availability of greater binding sites
around the surface of PAH film on quartz substrate and in course of time all the
interaction with CR molecules at all the binding sites gets over and no PAH remains free
for further interaction. Inset of figure 6 is a plot of the maximum absorbance as a
function of time. The experimental result (solid line) is found to fit well to the second
order kinetic rate model given by the equation [17].
![]()
Where A is the absorbance
(taken as proportional to the amount of adsorbed material), k1, k2
and n are constants.![]() and
and ![]() are the characteristic times. The dotted line in
the inset of figure 6 shows the curve generated by fitting to the above equation. Table 1
shows the values of different parameters used for fitting the experimental data.
are the characteristic times. The dotted line in
the inset of figure 6 shows the curve generated by fitting to the above equation. Table 1
shows the values of different parameters used for fitting the experimental data.
Table 1
| Parameters used in equation 1 for fitting Experimental data |
| k1 = 0.00996 |
| k2 = 0.00989 |
| n = 1.0 |
3.4 Effect of pH and adsorption
A remarkable dependence on pH of the CR
solution was found for PAH-CR films fabricated at different pH value of anionic solution.
Figure 7 shows the absorption spectra of PAH-CR films fabricated at different pH of
solution. It is observed that adsorption increased with the decrease of pH and decreased
with the increase of pH. The increase in adsorption of CR molecules may be due to
increased protonation of the amine group of NH3+ of PAH. CR is an
acidic dye and contains negatively charged SO3- group which
facilitates a higher adsorption of dye at lower pH due to the increase in electrostatic
attraction between negatively charged dye molecule and positively charged NH3+
of PAH surface. At high pH the charged surface of PAH films interacts with the
negative hydroxyl ions and thereby hinders the adsorption of CR molecules due to
electrostatic repulsion at the binding sites as CR is a negatively charged anionic dye.
Hydrogen bonding and Vanderwaals force may be responsible at higher pH of solution.
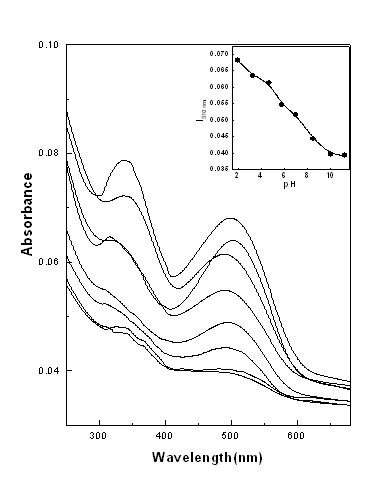
Fig. 7 The absorption spectra of PAH-CR films
fabricated at different pH of solution.
4. CONCLUSION
In summary, we have demonstrated
successful fabrication of ELBL self assembled films of PAH- CR without any material loss.
The layer effect study showed an increase in absorption intensity with the increase of
number of layers which confirmed the growth of film. The binding property study marked a
successful incorporation of CR molecules in the PAH-CR ELBL films. Thorough analysis of
UV-Vis absorption spectra and second derivative analysis revealed the formation of
molecular aggregates of CR molecules in PAH-CR ELBL self assembled films. The interaction
process between PAH and CR molecules was found to complete in 10 minutes and the
adsorption data were found to fit well to the second order adsorption model of describing
interaction mechanism. There was an inverse variation of adsorption of CR molecules with
the pH of the solution.
REFERENCES
[1] Muruganandham M and Swaminathan M 2004 Dye Pigment 62 269-275
[2] Decher G, 1997 Science 277 1232¨C1237.
[3] Ulman A, 1991 An Introduction to Ultrathin Organic Films, Boston, MA Academic Press
[4] Baur J W, Durstock M F, Taylor B E, Spray J R, Reulbach S, Chiang L Y, 2001 Synth.
Met. 121 1547
[5] Colvin V L, Schlamp M C, Alvistos A P, 1994 Nature 370 354
[6] Ferreira M, Onitsuja O, Fou AC, Hsieh B, Rubner MF, 1996,Mater Res. Symp. Proc. 413,
49
[7] Hong H, Davidov D, Avny Y, Chayet H, Farggi E Z, Neumann R, 1995, Adv.Mater. 7 846¨C849
[8] Brands C, Neyman P J , Guzy M, Shah S, Wang H, Gibson H W, VanCott K, Davis RM, Figura
C, Heflin J R, 2000, Polym. Mater. Sci. Eng. 83,219
[9] Cheung JH, Stockton B, Rubner MF, 1997, Macromolecules 30, 2712
[10] Cassagnean, T Fendler J H, 1999 J. Phys. Chem.B,103,1789
[11] Caruse,F, Mohwald, H 1999,J.Am.Chem. Soc.,121,6039
[12] Hussain SA, Dey D, Paul PK, Bhattacharjee D , Sinha S 2007 Chemical Physics Letters
450 49¨C54
[13] Das S, Pal A J, 2002,Langmuir 18 , 458¨C461
[14] Edwards R A, Woods R W, 1983, J chem. Phys., 87, 1329-37
[15] Agira K, Lvov Y, Kunitake T 1997, J.Am. Chem. Soc. 119, 2224-31
[16] Cooper T M, Stone M O, 1998, Langmuir 14 6662
[17] Raposo M, Pontes R S,Mattoso L H C, Olivera Jr. O N, 1997, Macromolecules 30,
6095-6101
[18] Kasha M, Rawls H R, Ashraf M, 1965,Pure Appl. Chem. 11, 371-92
[19] Moebius D, 1995, Adv Mater, 5, 437-444