|
Molbank 2005, M396 |
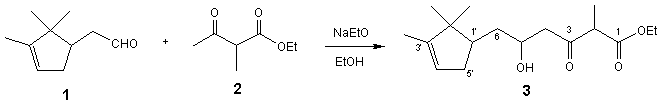
5-Hydroxy-2-methyl-3-oxo-6-(2,2,3-trimethyl-cyclopent-3-enyl)-hexanoic acid ethyl ester
Mercedes P¨¦rez-Bonilla, Juan M. Castro, Pablo J. Linares-Palomino, Sof¨Şa
Salido, Joaqu¨Şn Altarejos,* Manuel Nogueras and Adolfo S¨˘nchez
Departamento de Qu¨Şmica Inorg¨˘nica y Org¨˘nica, Facultad de Ciencias
Experimentales,
Universidad de Ja¨¦n, 23071 Ja¨¦n, Spain.
Tel.:
34-953-002743, Fax: 34-953-012141, e-mail: [email protected]
Received:
Keywords: a-Campholenic aldehyde, ethyl 2-methylacetoacetate, aldol
reaction.
Sodium (241 mg, 10.48 mmol) was added to
absolute ethanol (40 mL) and the mixture refluxed until
sodium reacted completely. Then ethyl 2-methylacetoacetate (2) (1.2 mL,
8.25 mmol) was added to the solution. The mixture was
refluxed for 1 h and, then, after reaching room temperature, aldehyde 1 (1.26
g, 8.27 mmol) was added and the mixture refluxed
again for 2 h. Finally, ethanol was partially evaporated under reduced pressure
and the residue neutralized with aqueous 2N HCl and
extracted with EtOAc (3ˇÁ15 mL).
The combined organic layers were washed with 2N HCl
(10 mL), saturated NaHCO3 (2ˇÁ15 mL) and brine (3ˇÁ10 mL). The
organic phase was dried over anhydrous Na2SO4 and the
solvent evaporated under reduced pressure to yield a residue (1.65 g) which was
purified by flash chromatography on silica gel, using a 6.5:4.5 hexane/EtOAc mixture as eluent, to give
the title compound 3 (356 mg, 1.20 mmol, 15%).
Melting point: 131.0¨C132.8 ºC
(white dust, from hexane/ethyl acetate).
IR (neat, cm-1):
3450¨C2600, 1101 (OH); 3036 (C=C); 1610 (CO); 1610, 1246, 1153 (COOEt).
1H NMR (300 MHz, CDCl3): ¦Ä= 0.79 (3H, s, Me-2ˇŻ); 1.01 (3H, s, MeˇŻ-2ˇŻ); 1.21 (3H, t, J=7.0 Hz, CH3CH2O), 1.36 (3H, d, J=6.6 Hz, Me-2); 1.62 (3H, br s, Me-3ˇŻ); 1.70¨C2.87 (7H, m, H-4, H-6, H-1ˇŻ, H-5ˇŻ); 3.48 (2H, q, J=7.0 Hz, CH3CH2O); 3.61 (1H, q, J=6.6 Hz, H-2); 4.69¨C4.81 (1H, m, H-5); 5.24 (1H, br s, H-4ˇŻ).
13C NMR (75 MHz, CDCl3): ¦Ä= 169.92 (C-1); 51.68 (C-2); 201.52 (C-3); 43.98 (C-4); 73.19 (C-5); 35.25* (C-6); 7.71 (Me-2); 15.22 (CH3CH2O); 65.81 (CH3CH2O); 44.91 (C-1ˇŻ); 46.79 (C-2ˇŻ); 148.67 (C-3ˇŻ); 121.00 (C-4ˇŻ); 35.17* (C-5ˇŻ); 19.73 (Me-2ˇŻ); 25.46 (MeˇŻ-2ˇŻ); 12.54 (Me-3ˇŻ).
*These signals may be interchanged.
EI-MS (70 eV, m/z): 279 (M+¨COH, 0.1%); 267 (M+¨CEt, 0.1);
251 (M+¨COEt, 0.8); 235 (0.7); 217 (0.9);
205 (0.4); 193 (1); 171 (2); 161 (9); 141 (9); 133 (11); 119 (20); 108 (C8H12+,
100); 93 (29); 79 (12); 43 (34).
Acknowledgements
We wish to thank the Ministerio de Ciencia y Tecnolog¨Şa for financial support (R+D Project
PPQ2000-1665) and the Ministerio de Educaci¨®n, Cultura y Deporte for a
fellowship to J. M. Castro.
© 2005 MDPI. All rights reserved.