|
Molbank 2005, M431 |
5-Methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine
Mariusz Mojzych* and Danuta Branowska
Institute of Chemistry University of Podlasie
ul. 3 Maja 54,
08-110 Siedlce, Poland
e-mail: [email protected]
Received:
Keywords: fluviols, nucleophilic
substitution.
O-
and N-alkyl derivatives of pyrazolo[4,3-e][1,2,4]triazine, isolated
from the cultural fluids of Pseudomonas fluorescens and Nostoc spongiaeforme exhibit antimicrobial and antitumor activity [1-2]. In this communication we report
the synthesis of 5-methoxy-1,3-dimethyl-1H-pyrazolo[4,3-e][1,2,4]triazine (2)
with the goal of performing structure-activity relationship studies.
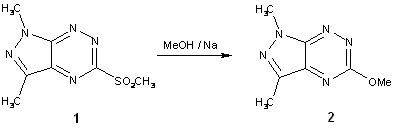
To a
solution of the sulfone 1 (145 mg, 0.5 mmol) in absolute methanol
(10 ml) metal sodium (35 mg, 1.5 mmol) was added. The
reaction mixture was heated at reflux for 30 min. After usual work-up compound 2 was purified by column chromatography
(Merck, silica gel 230-400 mesh) using CHCl3 as eluent.
The 1,3-dimethyl-5-methoxy-1H-pyrazolo[4,3-e][1,2,4]triazine (2) was isolated in 96% yield.
Melting Point: 110ºC.
1H-NMR (200 MHz, CDCl3):
δ= 2.59 (s, 3H); 4.20 (s, 3H); 4.24 (s, 3H).
IR (KBr, cm-1):
2960; 1310; 1120; 1050.
MS- EI (m/z):
179[M+].
Elemental Analysis: Calculated for C7H9N5O: C,
46.93%; H, 5.02%; N, 39.10%. Found: C, 47.20%; H, 4.94%; N, 38.89%.
References:
1.Smirnov, V.V.; Kiprianowa,
E.A.; Garagulya, A.D.; Esipov,
S. E.; Dovjenko, S. A., FEMS Microbiology Lett., 1997, 153, 357.
2.Hirata, K.; Nakagami, H.; Takashina, J.; Mahmud, T.; Kobayashi, M.; In, Y.; Ishida, T.; Miyamoto, K., Heterocycles, 1996, 43, 1513.
Sample Availability: Available from MDPI.
© 2005 MDPI. All rights reserved.