Electrophilic Substitution at
C(7) of a Protected 7-Deaza-2’-deoxyguanosine –
The
2’-Deoxyribonucleoside Parent Analogue of Queuosine #
Natalya Ramzaeva and Helmut
Rosemeyer *
Organische Chemie I –
Bioorganische Chemie, Institut für Chemie, Fachbereich Biologie/Chemie,
Universität Osnabrück, Barbarastr. 7, D-49069 Osnabrück, Germany
*
Author to whom correspondence should be addressed. E-Mail: [email protected]
Received: 28 March 2007 /
Accepted: 4 April 2007 / Published: 23 April 2007
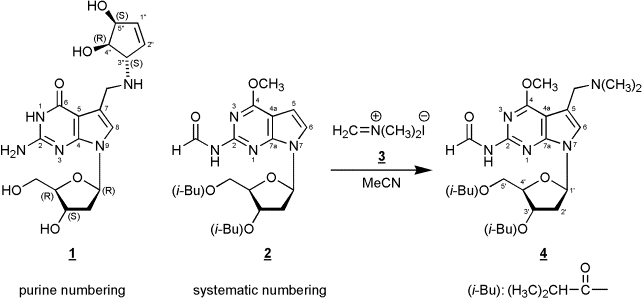
Abstract: In this manuscript we
report on the regioselective electrophilic substitution at C(7) of
7-[2-deoxy-3,5-bis-O-(2-methylpropanoyl)-ß-D-erythro-pentofuranosyl]-2-(formylamino)-4-methoxy-7H-pyrrolo[2,3-d]pyrimidine
(2) – a
protected precursor of 7-deaza-2’-deoxyguanosine - using
N,N-dimethyl-methyleniminium
iodide (Eschenmoser’s salt) yielding the Mannich compound 4.
Keywords: 7-Deazapurine, Mannich
bases, Eschenmoser’s Salt
Most naturally occurring 7-deazapurine
ribonucleosides [1,2]
– both of the adenosine as well as of the guanosine type – carry
substituents at the 7-position, and many of them which are found in
tRNA represent Mannich bases such as the nucleoside
“Q” {2-amino-5-(4,5-cis-dihydroxy-1-cyclopenten-3-yl-trans-aminomethyl-(7-ß-D-ribofuranosyl)-3,7-dihydro-4H-pyrrolo[2,3-d]pyrimidin-4-one,
Queuosine, 1}. Many attempts have been made to direct a Mannich side
chain regioselectively into position 7. In contrast to 7-deazaadenosine
derivatives [3,4] this is not an easy task for
7-deazaguanosine precursors [5]
and requires chemical detours [6].
For example, regioselective C(7) Mannich alkylation (morpholine,
HCOH/HOAc) can be performed at 3’,5’-bis-O-toluoylated
6-methoxy-2-(methylthio)-7-deazapurine 2’-deoxy-ß-D-ribonucleoside,
followed by a three-step aglycone conversion to 7-deazaguanine and
deprotection of the glycone [7].
It has been reported for
7-deazaguanines that the position of electrophilic substitution
strongly
depends on the particular substituent pattern of the base as well as of
the
reaction conditions [8-10]. Of
decisive importance is the observation that a
free 2-amino group directs the electrophilic attack into the undesired
8-position (position 6 using systematic numbering) of the
7-deazaguanine
moiety. This is the result of mesomeric stabilization of the σ-complex
formed
during electrophilic attack at the 8-position. On the other hand,
2-acylamino-7-deazaguanine derivatives form the desired 7-substituted
compounds
[8]
In this communication we
disclose
that reaction of the fully protected 7-deaza-2’-deoxy-7-deazaguanosine
derivative 2 [8]
with
N,N-dimethyl-methyleniminium iodide (Eschenmoser’s salt) afforded the
C(7)
alkylated Mannich compound 4 in
moderate yield. This reaction presents a new and alternative route to
7-deazapurine nucleosides with a Mannich side chain in position 7. The
structure of 4 was unequivocally
assigned by 1H- and 13C-NMR
spectroscopy as well as by 1H-NMR
NOE difference spectroscopy (see Experimental Procedure). 13C-NMR
resonances were assigned applying DEPT-135 and [C,H]HETCOR spectra.
A further
transformation of the tertiary amino
group of 4 into other Mannich
compounds can be accomplished after quarternization. Furthermore, the
synthetic
or enzymatic incorporation of Mannich bases derived from compounds such
as
compound 4 allows the introduction
of reporter groups in a favourable position of a DNA molecule because
the
Mannich side chain protrudes into the major groove of a B-DNA double
helix.
Experimental Procedure
7-[2-Deoxy-3,5-bis-O-(2-methylpropanoyl)-ß-D-erythro-pentofuranosyl]-5-(dimethylaminomethyl)-2-(formylamino)-4-methoxy-7H-pyrrolo[2,3-d]pyrimidine
(4).
Compound 2 (305 mg, 0.7 mmol) was dissolved in
acetonitrile (3 mL) and N,N-dimethyl-methyleniminium iodide (3, Eschenmoser’s Salz, 260 mg, 1.4
mmol) was added. After heating to 80°C for 24h, the reaction mixture
was
evaporated to a small volume, and compound 4
(67 mg, 20 %) was isolated as an amorphous solid by thick-layer
chromatography
(chloroform/methanol, 9:1, two developments, Rf,
0.4), elution from
the silica gel with methanol and centrifugation. 1H-NMR ((D6)DMSO,
500.1 MHz): δ 10.73 (1H, d, HC=O,
J = 9.9 Hz); 9.44 (1H, d, NH, J = 9.3 Hz); 7.31 (1H, s,
H-C(6)); 6.49 (1H, dd, H-C(1’), J = 6.4, 8.0 Hz); 5.36 (1H, dd,
H-C(3’), J =
2.8, 3.6 Hz); 4.28 - 4.17 (3H, m, H-C(4’), H2-C(5’));
4.03 (3H, s,
OCH3); 3.67 (2H, s, CH2);
2.86 (1H, m, iBu-CH); 2.63 –
2.57 (2H, m, H2-C(2’)); 2.47 (1H, m, iBu-CH);
2.28 (6H, s, N(CH3)2);
1.15 and 1.09 (12H, 4 iBu-CH3). 1H-NMR
NOE Data ((D6)DMSO):
proton irradiated: H-C(1’). NOE observed: H-C(6), 1.5 %; Hα-C(2’),
2.5 %; H-C(4’), 1.5 %. 13C-NMR
((D6)DMSO, 125.8 MHz): δ 175.6, 175.8 (2 C=O,
iBu); 163.4 (C-4);
163.3 (C=O, formyl); 152.4 (C-7a); 152.0 (C-2); 121.6 (C-6); 111.4
(C-5); 101.5
(C-4a); 82.9 (C-1’); 81.1 (C-4’); 74.2 (C-3’); 63.6 (C-5’); 53.8 (CH2);
53.7 (OCH3); 44.1 and 33.1 (4 Me, iBu); 35.9
(C-2’); 18.6 (2 Me,
N(CH3)2).
References and Notes
- Suhadolnik,
R. J., Nucleosides As Biological Probes.
John Wiley & Sons, New York 1979,
pp. 158-169.
- Limbach,
P. A.; Crain, P. F.; McCloskey, J. A. Nucleic
Acids Res. 1994, 22, 2183.
- Watanabe,
S. I.; Ueda, T. Nucleosides Nucleotides
1983, 2,
113.
- West,
R. A. J. Org. Chem. 1961,
26, 4959.
- Seela,
F. Lüpke, U. Chem. Ber. 1977, 110,
1462.
- Seela, F.; Richter, R. Chem.
Ber. 1978, 111,
2925.
- Seela, F.; Chen, Y.; Zulauf, M. Synthesis
1997, 1067.
- Ramzaeva,
N.; Seela, F. Helv. Chim. Acta 1995, 78,
1083.
- Akimoto,
H.;
Imamiya, E.; Hitaka, T.; Nomura, H.; Nishimura, S. J.
Chem. Soc. Perkin Trans 1
1988, 1637.
- Benghiat, E.;
Crooks, P. A. J.
Heterocycl. Chem. 1983, 20, 1023.
#
Purine numbering has been used within the General Part of the
manuscript, systematic numbering within the Experimental Part.
©
2007 by MDPI (http://www.mdpi.org/).
Reproduction is permitted for noncommercial purposes.

