|
Molbank 2007, M531 |
Synthesis of 2-[4-(2-chlorobenzyl)-3-methyl-6-oxopyridazin-1(6H)-yl]acetohydrazide
Nour-Eddine Benchat *, Abderrahmane
Anaflous, Said Abouricha, Brahim El-Bali and Mohamed Ramdani
Department of Chemistry, University
Mohamed I, Sciences Faculty, 60000
*Author to whom correspondence should be addressed. E-mail: [email protected]
Received: 3 January 2006 / Accepted: 24 June 2006 / Published: 31 May 2007
Keywords:
pyridazin-3(2H)-one, N-alkylation, anticonvulsive agents.
Pyridazines are of chemical and biological interest.
They have been reported to be anticonvulsive agents [1], [2]. Furthermore, BELLASIO
et al. have described the
antihypertensive effects of hydrazinopyridazine
compounds [3]. In continuation of this line of investigation, we have
synthesized compound (I); it
will be subjected to further pharmacological investigations, especially tests
of its anticancer activity.
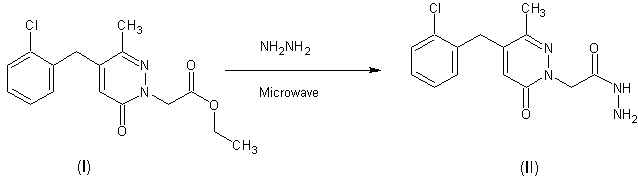
To (
Melting point: 201-
IR (KBr,
cm-1): 3352(NH), 1682 , 1469 , 1622 (C = O)
1HNMR (300.14 MHz, CDCl3) d (ppm): 2.49 (s, 3H,
CH3), 3.97 (s, 2H, CH2), 4.21 (s, 2H, NH2),
4.52 (s, 2H, CH2), 6.03 (s, 1H, H4), 7.53 (m, 4H, H aromatic), 9.24
(s, 2H, NH2).
13CNMR (75 MHz, CDCl3) d (ppm): 19.38 (CH3),
35.6 (CH2), 52.76 (CH2), 126.98 (CH aromatic), 128.85 (CH
aromatic), 130.25 (CH aromatic), 130.68 (CH aromatic), 132.6 (CH aromatic),
134.54, 135.43, 144.93, 146.03, 160.31 (C=O), 167.16 (C=O).
References:
1. Wermuth, C.G.; Leclerc, G.; Melounov, P. Chim. Ther. 1971, 2, 109.
2. Laborit, H.; Weber, B.; Wermuth,
C.G.; Delbarre, B.; Chekler,
C.; Baron, C.; Rosen Garten, H. AGRESSOLOGIE 1965, 6, 415.
3. Bellasio, E. ; Parravicini, F. ; Testa,
4.
(a) Kappe,
C.O.; Dallinger, D. Nature Reviews Drug Discovery 2006, 5, 51; (b) De
© 2007 by MDPI (http://www.mdpi.org/). Reproduction is permitted for noncommercial purposes.