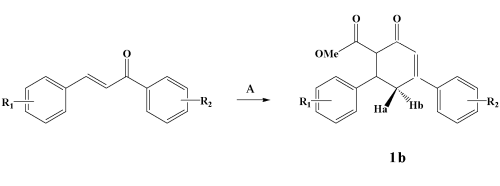
Scheme 1. Chemical synthesis of methyl 1-oxo-3,5-diphenylcyclohexene-6-carboxylate 1b (R1= R2: H). A represents CH3COCH2CO2CH3, NaOMe, MeOH, rt.
| Molbank 2007, M558 | http://www.mdpi.org/molbank/ |
Received: 8 May 2007 / Accepted: 4 September 2007 / Published: 19 November 2007
Introduction
The exploration of a simple molecule with different functionalities for the synthesis of heterocycles is a worthwhile contribution in the chemistry of heterocycles. In fact, 6-ethoxycarbonyl-3,5-diarylcyclohexenone has been used as an effective synthon in some projected syntheses of benzoselenadiazoles/thiadiazoles1, spirocyclohexanones2, carbazole derivatives3, fused isoxazoles and pyrazoles4,5. The intermediate present in this communication has been chosen by us as a promising starting material to develop pyrazole rings with antimalarial and antibacterial activities6.Compounds 1a,b were synthesised by a base-catalysed
Claisen-Schmidt condensation of 4-chlorobenzaldehyde with the appropriate
acetophenone. These ketones on treatment with methyl 3-oxobutanoate in the presence
of sodium methoxide yield methyl 6-(4-chlorophenyl)-4-(4-methylphenyl)cyclohex-3-en-2-one-1-carboxylate7
1a (R1: 4-Cl; R2: 4-Me in
Scheme 1) or methyl-1-oxo-3,5-diphenylcyclohexene-6-carboxylate
1b (R1= R2: H in Scheme 1, see also Figure 1). In the present communication,
we report the formation of 1b and
its characterisation by elemental analysis as well as IR, 1H NMR, 13C
NMR, COSY, and HETCOR spectral and crystallographic studies. The analysis of 1a has been previously reported7.

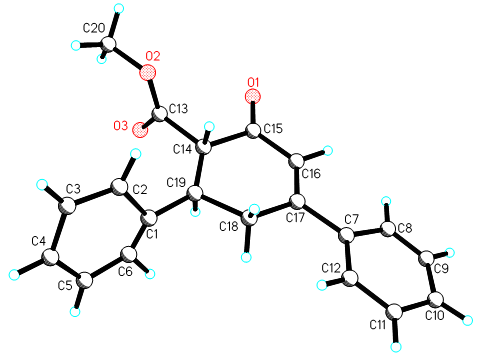
Figure 1. View of the structure of 1b with the atom numbering for carbon and oxygen atoms, showing
displacement ellipsoids at the 50% probability level.
Melting points were
determined on a Thomas micro hot stage apparatus and are uncorrected. Infrared
spectra were determined as KBr pellets on a Shimadzu model 470
spectrophotometer. The 1H NMR, 13C NMR, COSY and HETCOR
spectra were recorded using a Jeol Eclipse 270 MHz spectrometer. Chemical
shifts are expressed relative to residual chloroform. Central Service of
Universidad de Málaga, Málaga-España performed elemental analyses, results were
within ± 0.4% of predicted values for all compounds. Chemical reagents were
obtained from Aldrich Chemical Co.,
X-ray Crystallographic Data Collection and Structural Determination.
Single-crystal X-ray
diffraction measurement of the compounds were carried out with a Bruker Smart
1000 CCD diffractometer equipped with a graphite crystal monochromator situated
in the incident beam for data collection at 298(2) K. The lattice parameters were obtained by
least-squares refinement of the diffraction data of 9336 reflections, and data
collections were performed with Mo Kαradiation (λ= 0.71073 Å) by ω scan mode in
the range of 1.96 < θ < 25.10°. All of the measured
independent reflections were used in the structural analysis, and semiempirical
absorption corrections were applied using the SADABS program. The maximum and
minimum transmission factors were 0.980 and 0.961. The program
SAINT14 was used for integration of the diffraction profiles. The structure was
solved by direct methods using the SHELXS97 program of the SHELXTL package and
refined with SHELXL9715. The non-hydrogen atoms were located in successive
difference Fourier syntheses. The final refinement was performed by full-matrix
least-squares methods with anisotropic thermal parameters for all of the
non-hydrogen atoms on F2. All of the
hydrogen atoms were generated theoretically onto the specific atoms and refined
isotropically with fixed thermal factors. A summary of the crystallographic
data and structure refinement of 1b
is listed in Table 1, and selected bond lengths and bond angles are listed in
Table 2.
Table 1. Crystallographic data for compound 1b
|
Parameter |
Compound
1b |
Parameter |
Compound
1b |
|
formula fw lattice a, Å b, Å c, Å β, deg V, Å3 space group Z |
C20
H18 O3 306.34 Monoclinic 14.885(7) 11.172(5) 9.367(4) 101.161(12) 1528.2(12) P2(1)/c 4 |
ρ(calcd), g cm-3 F(000) μ(Mo KR), cm-1 diffractometer radiatn λ, Å temp, ْ C R (I > 2.00σ(I))a Rw (all data)b no. of observs no. of variables |
1.331 648 0.089 APEX 0.71073 25 0.3288 0.6053 2671 (all data) 210 |
Table 2. Selected bond distances (Å), angles (deg) and
torsion angles (deg) for compound 1b
at 298 K
|
Distances |
|
C(15)-O(1) 1.20(2) C(15)-C(16) 1.41(2) C(15)-C(14) 1.52(3) C(13)-O(3) 1.18(3) C(13)-O(2) 1.24(3) C(13)-C(14) 1.50(3) C(1)-C(6) 1.04(3) C(1)-C(19) 1.36(3) C(1)-C(2) 1.49(3) C(7)-C(12) 1.34(3) C(7)-C(8) 1.37(3) C(7)-C(17) 1.50(3) C(8)-C(9) 1.24(3) |
|
angles |
|
O(1)-C(15)-C(16) 126.9(19) O(1)-C(15)-C(14) 121.1(16) C(16)-C(15)-C(14) 112.0(18) O(3)-C(13)-O(2) 127(3) O(3)-C(13)-C(14) 116(3) O(2)-C(13)-C(14) 117(3) |
|
torsion angles |
|
C(12)-C(7)-C(8)-C(9)
3(3) C(17)-C(7)-C(8)-C(9) -174(2) C(7)-C(8)-C(9)-C(10) -4(4) C(8)-C(9)-C(10)-C(11) 7(4) C(9)-C(10)-C(11)-C(12) -10(4) C(8)-C(7)-C(12)-C(11) -4(3) C(17)-C(7)-C(12)-C(11) 172.4(19) C(10)-C(11)-C(12)-C(7) 8(3) O(1)-C(15)-C(16)-C(17) -173(3) C(14)-C(15)-C(16)-C(17) 8(4) C(15)-C(16)-C(17)-C(18) -10(3) C(15)-C(16)-C(17)-C(7) 174(2) C(12)-C(7)-C(17)-C(16) 168(2) C(8)-C(7)-C(17)-C(16) -15(3) C(12)-C(7)-C(17)-C(18) -7(3) C(8)-C(7)-C(17)-C(18) 169.5(19) C(16)-C(17)-C(18)-C(19) 21(3) C(7)-C(17)-C(18)-C(19) -163.7(18) C(6)-C(1)-C(19)-C(14) 127(4) C(2)-C(1)-C(19)-C(14) -58(4) C(6)-C(1)-C(19)-C(18) -75(3) C(2)-C(1)-C(19)-C(18) 100.6(19) C(17)-C(18)-C(19)-C(14) -29(3) C(17)-C(18)-C(19)-C(1) 167.5(19) O(3)-C(13)-O(2)-C(20) -2(5) C(14)-C(13)-O(2)-C(20) -179(3) C(19)-C(1)-C(6)-C(5) -175(3) C(2)-C(1)-C(6)-C(5) 10(4) |