|
Molbank 2006, M455 |
Synthesis
and Physical Characterization of 2-((E)-1-(3-((E)-1-(2-hydroxyphenyl)ethylideneamino)-2-methylphenylimino)ethyl)phenol
A. A. Jarrahpoura*, A. F. Jalboutb*,
S. Rezaeia and B. Trzaskowskib
aDepartment of Chemistry,
bDepartment of Chemistry,
Phone: +98 711 2284822,
Fax: +98 711 2280926, e-mail: [email protected] or [email protected]
*Author
to whom correspondence should be addressed
Received:
Abstract: In this paper we propose the synthesis of
2-((E)-1-(3-((E)-1-(2-hydroxyphenyl) ethylideneamino)-2-methylphenylimino)
ethyl) phenol. In addition to its synthesis we present AM1 and B3LYP/6-31G*
calculations to characterize the physical properties of the molecule.
Keywords: 2-Hydroxyacetophenone, 2-methyl-1,3-phenylenediamine, Schiff base, AM1, B3LYP
Introduction:
Schiff bases are important intermediates for the synthesis of various bioactive compounds [1-2]. Furthermore, they are reported to show a variety of biological activities including antibacterial [3], antifungal [4], anti mouse hepatitis virus (MHV) [5], inhibition of herpes simplex virus type 1 (HSV-1) and adenovirus type 5 (Ad 5 )[6], anti cancer [7], anti mosquito larvae [8] and herbicidal activities [9].The complexes containing the nontoxic 2-hydroxyacetophenone have been used in selective membrane electrodes[10].Choudhuri et al have synthesized a copper complex of 2-hydroxyacetophenone and they have evaluated it as an anticancer agent [11].Some Co(III) complexes of 2-hydroxyacetophenone have been synthesized by John and his coworkers[12].Grunule group have synthesized and characterized four copolymer derived from 2-hydroxyacetophenone.In view of these facts we decided to synthesize a new Schiff base from the nontoxic 2-hydroxyacetophenone as potential biological and complexometric agent. Its biological activities and analytical works are under study.
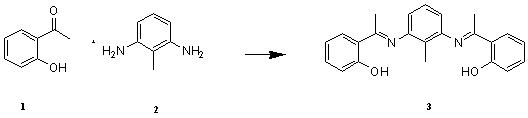
Results and Discussion:
2-Hydroxyacetophenone 1 (2.03 g, 1.8 mL,
15 mmol) and 2-methyl-1, 3 pheneylenediamine
2 (0.61 g, 5 mmol)
were dissolved in 20 ml of warm ethanol. The
reaction mixture was refluxed for 8h at 85 ��C, and allowed to stand. The solid
crystals were filtered off and washed with ethanol. The pure Schiff base 3
was isolated as a light yellow crystalline solid (yield 68%).We next performed theoretical calculations to
present a viable structure for the product. All calculations in this work where
carried out with the AM1 level of theory using the GAUSSIAN 03 [13] suite of
programs. More information about these methods is available elsewhere [14].
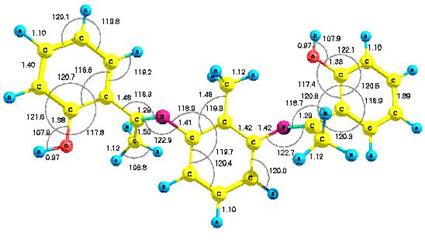
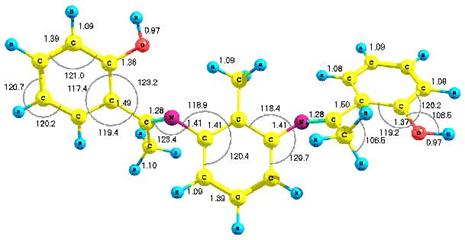
Figure 1 presents the optimized structure of the molecule with bond lengths and
bond angles shown. We obtained a
melting point (mp) value 184-186 ��C, and IR (KBr, cm-1):
3244(OH) (B3LYP/6-31G*: 3217); 1604(C=N) (B3LYP/6-31G*: 1629), as well as NMR.
|
|
|
|
(A) |
(B) |
|
Figure 1. (A). AM1 optimized geometry and (B) B3LYP/6-31G* optimized
geometry with all bond lengths shown in angstroms (Å), and bond angles in
degrees (º) |
|
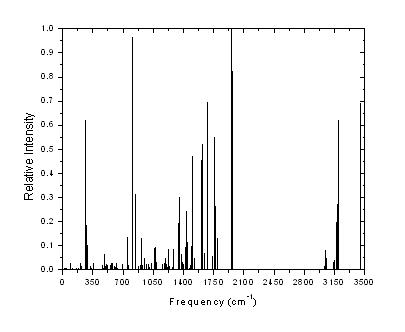
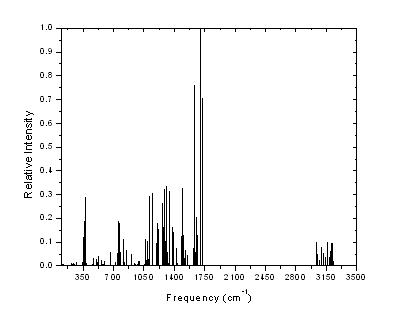
Figure 2 shows the theoretical IR vibrational spectrum for this molecule.
|
|
|
|
(A) |
(B) |
|
Figure 2. (A). AM1 IR Spectra theoretical, (B) B3LYP/6-31G* theoretical IR Spectra |
|
Table 1 shows the thermodynamic properties for the complex in figure
1 where T (temperature in K), S (entropy in J mol-1 K-1),
Cp (heat capacity at constant pressure in kJ mol-1 K-1),
and ��H=H�� - H��298.15 (enthalpy content, in kJ mol-1),
T1=100 K, T2=298.15 K, and T3=1000 K calculated
AM1 frequencies. The fits were performed according to the equations implemented
by the National Institute of Standards and Technology (NIST) [15].
|
|
|
Fitted Thermodynamic
Equation (T/1000=t) |
100 K |
298.15K |
1000
K
|
|
AM1 |
Cp |
-32.60241+ 1692.63626*t -907.85165*t2 + 150.38779*t3 +0.51679*t-2 |
179.04 |
398.44 |
904.39 |
|
S |
53.732
*ln(t) + 1197.55633 *t + 17.76292 *t2/2 -370.35669 *t3/3
- 5533.6327 /(2*t2) + 153.29627 |
462.67 |
755.21 |
1541.55 |
|
|
��H |
465.16043
*t + 6885.20409 *t2/2 -13920.11871 *t3/3 + 7543.03025
*t4/4 �C 7.7704 /t -1756.97223 |
11.50 |
68.42 |
560.53 |
|
|
B3LYP/6-31G* |
Cp |
-72.8544+ 1979.22049*t -1324.98024*t2 + 333.74311*t3 +0.56512*t-2 |
168.36 |
412.38 |
916.56 |
|
S |
35.62635*ln(t)
+ 1302.38545*t + 32.60854*t2/2 -463.77548*t3/3 +
2344.59164/(2*t2) + 159.49792 |
439.53 |
731.65 |
1540.23 |
|
|
��H |
-101.60395*t
+ 6628.71227*t2/2 -13286.08662*t3/3 + 7109.30838*t4/4
+ 14.26015/t +702.59222 |
10.54 |
67.85 |
572.53 |
Table 1. Thermodynamic properties of the molecule in Figure 1, calculated at the AM1 level and B3LYP/6-31G* level of theory, where Cp is the heat capacity in J mol-1 K-1, S is the entropy in J mol-1 K-1, and DH is the standard enthalpy kJ mol-1. These where fitted to the Shomate equations [15] which are implemented by the JANAF tables of the NIST databases. These equations converged to an R2 value of 0.999 on average.
These equations have been very good
at predicting physical properties of various molecules, as we have tested in
the past [16-19]. Overall, there is some relative correlation between the AM1
and B3LYP/6-31G* values, however, the density functional theory values should
be much more reliable.
Melting Point: 184-186 ��C
IR (KBr, cm-1): 3244(OH); 1604(C=N).
1H-NMR
(250 MHz, CDCl3): 1.65(6H, s, ArCH3), 2.26(3H, s, ArCH3),
6.21(2H, d, Ar), 6.24(2H, d, Ar),
6.88-7.64(5H, m, Ar), 7.94(2H, d, Ar),
14.57(2H, s, OH).
13C-NMR
(62.9 MHz,CDCl3):
11.516; 17.133; 30.944; 111.21; 111.71; 113.20; 118.02; 118.22; 119.63;
126.66; 128.88; 132.95; 145.55;145.60; 146.60; 162.15; 171.23.
MS
(m/z): 358.47, 225, 132, 106, 77.
Acknowledgment
AFJ and BT would like to
thank the
References:
1. a) Jarrahpour, A. A.; Shekarriz, M. and Taslimi, A. Molecules 2004, 9, 29-38 b) Hakimelahi, G. H.; Jarrahpour, A. A. Helv. Chim Acta. 72(7), (1989), 1501-5.c) Hakimelahi,G. H.; Jarrahpour,A. A. J. Sci. R. Iran 1(5) (1990) 353-354. d) Venturini, A.; Gonzalez, J. J. Org. Chem. 2002, 67, 9089.
2. Taggi, A. E.; Hafez, A. M.; Wack, H.; Young, B.; Ferraris, D.; Lectka, T. J. Am. Chem. Soc. 2002, 124, 6626.
3. JaEl-masry, A. H.; Fahmy, H. H.; Abdelwahed, S. H. A. Molecules 2000, 5, 1429.
4. Singh, W. M.; Dash, B. C. Pesticides 1988, 22(11), 33.
5. Wang, P.H.;
Keck, J. G.; Lien, E. J.; Lai, M. M. C. J. Med. Chem. 1990, 33
(2), 608.
6. Das, A.;
Trousdale, M. D.; Ren, S.; Lien, E. J. Antiviral Res. 1999, 44(3), 201.
7. Desai, S. B.;
Desai, P. B.; Desai, K. R. Hetrocycl. Commun. 2001, 7(1), 83.
8. Das, B. P.; Choudhury, R. T.; Das, K. G.; Choudhury, D. N.; Choudhury, B. Chem. Environ. Res. 1994, 3 (1&2), 19.
9. Samadhiya, S.; Halve, A. Orient. J .Chem. 2001, 17 (1), 119.
10. Mazloum Ardakani, M.; Salavati-Niasari, M.; Jamshidpoor, M. Sensors and Actuators 2004, B101, 302.
11. Majumder, S.;
Panda , G. S.; Choudhuri, S. K. Eur. J. Med. Chem. 2003, 38, 893
12. John, R. P.;
Sreekanth, A.; Kurup M. R. P.; Mobin, S. M. Polyhedron 2002, 21, 2515-/2521.
13. Frisch, M.J.,
et.al., GAUSSIAN 03, Revision A.1, M. J. Frisch, et. Al., Gaussian, Inc.,
14. Foresman, J.B., Æ Frisch, Exploring Chemistry with Electronic Structure Methods, 2nd edition Gaussian, INC, Pittsburgh, PA, 1996
15. Linstrom, P.J., Mallard, W.G.,
Eds., NIST Chemistry WebBook, NIST Standard Reference Database Number 69, July 2001, National Institute of
Standards and Technology, Gaithersburg, MD 20899
16. Jalbout, A.F.
, Solimannejad, M., Labonowski,
J.K., Chem. Phys. Letts., 2003, 379,
503.
17. Jalbout, A.F., Jiang, Z.-Y., Quasri, A., Jeghnou, H., Rhandour, A., Dhamelincourt, M.C., Dhamelincourt, P., Mazzah, A., Vib. Spect., 2003, 33, 21.
18. Jalbout, A.F., Nazara, F., Turker, L., J.
Mol. Struct. (THEOCHEM), 2004, 627, 1. (Invited
Review)
19. Jalbout, A.F., Adamowicz, L., Solimmanejad, M., Chem.
Phys. Letts., 2006, xx-xx
Sample Availability: Available from MDPI
© 2006 MDPI. All rights reserved.