|
Molbank 2006, M487 |
2-[N-(O-ethylaminophenyl)
phenyl imidoyl methylylidene]-3,5-diphenyl-1,3,4-thiadiazole
J. Nathantene Ghomsi, M.Lamine Doumbia, Noureddine Hamou Ahabchane and El Mokhtar Essassi*
Laboratoire de Chimie Organique Hétérocyclique, Faculté des Sciences, Avenue Ibn Batouta, BP-1014-Rabat-Maroc.
e-mail : [email protected]
*Author to whom correspondence should be addressed
Received: 18 July 2006 /
Accepted: 20 July 2006 / Published: 1 September 2006
Keywords: cycladdition, benzodiazepine-2-thione, 1,3,4-thiadiazole.
4-phenyl-1,5-benzodiazepine-2-thione are used as starting materials in the synthesis of several heterocyclic compounds for potential biological actvities.1-3 The synthesis of a new 1,3,4-thiadiazole derivative is reported.
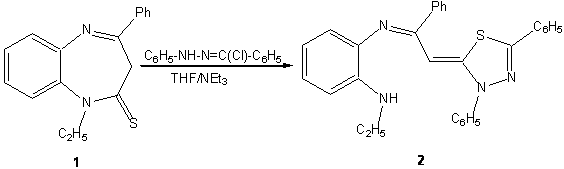
Scheme 1
To a
solution of 0.01mol of 1-ethyl-4-phenyl-1,5-benzodiazepine-2-thione
1 [1] in 60 ml of tetrahydrofuran, 0,02 mol of
hydrazonoyl chloride and 0.04 mol of triethylamine were added. Then, the mixture was refluxed
for 12 hours. After cooling, salts are removed by filtration and solvent
was evaporated under reduced pressure. The residue isolated was recrystallized from ethanol. The 1,3,4-thiadiazole
derivative 2 was obtained in 60 % yield.
Melting point: 165 °C
1H-NMR (CDCl3, 250 MHz ): δ= 1,34 (t, J = 7,12 Hz, 3H), 3,36 (q,
J = 7,12 Hz, 2H), 7,24 (s, 1H), 6,05-7,92 (m, 19H).
13C-NMR (CDCl3, 67.5 MHz ): δ= 15,24 (CH3), 38,94 (CH2),
90,19 (CH=), 110,00-130,26 (CHar), 130,53-142,35
(Car), 151,53 (C=N), 162,98 (C=N).
MS (EI): 474 [M]+
Elemental analysis: Calculated for C30H26N4S : C, 75.92%; H, 5.52%; N, 11.80%. Found: C, 75.97%; H, 5.46%; N, 11.84%;
References:
1. Essassi E. M. and
2. Essassi E. M., Bull. Soc. Chim. Belg., 1994, 103, 679.
3.
© 2006 MDPI. All rights reserved.