|
Molbank 2006, M510 |
Synthesis of
2-Chloro-3-(2-naphthyloxy)quinoxaline
Craig A. Obafemi*1,
Wolfgang Pfleiderer2
1Department of Chemistry,
2Fachbereich Chemie,
Konstanz University D-78457 Konstanz/ Germany
E-mail:
[email protected]
([email protected])
*Author to whom
correspondence should be addressed
Received: 7
February 2005 / Accepted: 2 February 2006 / Published: 1 December 2006
Keywords: 2,3-Dichoroquinoxaline, 2-naphthol.
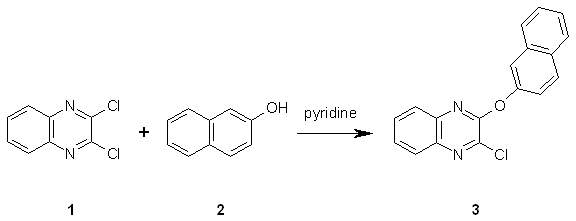
A mixture of 2,3-dichloroquinoxaline 1 [1] (1.0 g, 5 mmol) and 2-naphthol (0.72 g, 5 mmol) in pyridine (10 mL) was heated to 130¡ãC for 3 h, with magnetic stirring, to give a dark-brown reaction mixture. The reaction mixture was allowed to cool and then poured into cold water (50 mL). The resulting solid was filtered, dried and purified by column chromatography on silica gel, eluting with petroleum ether (b.p. 100¡ã) / ethanol (100:1) to afford 2-chloro-3-(2-naphthyloxy)quinoxaline 3 as colorless crystals (0.9 g, 58.0%).
Melting point: 163 ¨C 165¡ãC
1H-NMR (400 MHz, DMSO-d6, d (ppm): 8.10 ¨C 7.90 (m, 5H, Ar-H), 7.76 ¨C 7.67 (m, 3H, Ar-H), 7.62 ¨C 7.54 (m, 3H, Ar-H).
13C-NMR (100 MHz, DMSO-d6, d (ppm): 152.8, 150.1, 138.8, 138.6, 133.5, 131.0, 130.8, 129.6, 128.5, 127.7, 127.6, 127.4, 126.8, 126.6, 125.8, 121.5, 118.1.
MS (m/z, %): 308 ([M + 2]+ 31.8), 306 (M+, 100), 278 ([M-CO] +,19.0), 271 ([M-Cl]+, 23.6), 243 ([M-CO-Cl]+, 34.6), 163 ([M-C10H7O] +, 8.6), 127 (17.3), 115 (50.3), 102 (45.6), 90 (12.0), 75 (22.2).
Acknowledgment
We thank the Alexander von Humboldt Foundation for a post-doctoral fellowship (C.A.O.).
Reference
1. C.A. Obafemi and Wolfgang Pfleiderer, Helv. Chim. Acta 1994, 77, 1549 ¨C 1556.
© 2006 MDPI. All rights reserved.