|
Molbank 2006, M513 |
Synthesis of 1-benzyl-3-chloro-4-(3,4,5-trimethoxyphenyl)azetidin-2-one
Zhaoqi Yang*, Pinhua Sun
Institute of New Drug Research,
e-mail: [email protected]
* Author
to whom correspondence should be addressed
Received: 6 July 2006 / Accepted: 3
August 2006 / Published: 1 December 2006
Keywords: Combretastatin
A-4, Synthesis, analogue
Abstract
Combretastatin A-4, isolated from a South African tree Combretum caffrum, is one of the most potent anti-tomour agents. In this paper, an analogue of Combretastatin A-4 was synthesized in order to improve the anti-tumour activity. Compound was characterized by IR and 1HNMR.
Introduction
Cancer is the second major cause of death in the world. Although many researches about cancer all around the world, researchers still have no way to cure for most forms of human cancer. One common characteristic for all types of cancer is the requirement of a suitable blood supply. Tumor vasculature is an important and rapidly emerging target for anticancer therapy. Therefore, vasculature of tumor is nowadays intensively preclinically and clinically investigated as a new potential target for anti-tumor therapeutic strategy. [1-4]
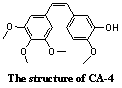
Combretastatin A-4 (CA-4), a natural
compound isolated from the bark of the South African bush willow tree Combretum caffrum (Combretaceae), is one of the most potent anti-vascular agents.
[5]
It
has also been demonstrated to shut down tumor vasculature rapidly, whereas the blood flow to normal
tissues was much less affected. [6-7] CA-4 has
received a great deal of attention due to its relatively simple structure, high
potency as cytotoxic agents and antivascular
activity. A number of CA-4 analogues has been synthesized and evaluated. [6] β-lactam is a very useful pharmacophore existed in a lot of drugs and Banik et al. [8] reported synthesis of β-lactam analogues entry to
novel anticancer agents. In
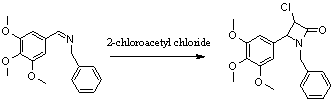
view of these facts, a new analogue of CA-4 is synthesized in this paper. The
biological evaluation of the compound would be done quickly.
Experimental
Melting points were uncorrected and were
measured with micro-melting point apparatus XT-4. IR spectra (KBr) were obtained on a Thermo Nicolet Nexus
Phenyl-N-(3,4,5-trimethoxybenzylidene)
methanamine (2 g, 7mmol) and triethylamine (3.9ml, 28mmol) dissolved in 50ml
anhydrous DCM, and the solution was stirred in an ice-salt bath under argon
atmosphere. The mixture was stirred for a few minutes, and then a solution of
2-chloroacetyl chloride (1.7ml, 21mmol) in 20ml DCM was added dropwise, and the reaction mixture was kept in ice-salt
bath stirred 24 hours. Diluted hydrochloric acid was added and stirred 5 minutes,
and then washed with 3×30ml saturated NaHCO3, 3×30ml saturated
brine, 40×2ml water. The organic layer was dried with anhydrous Na2SO4
and concentrated under reduced pressure. The residual was purified by
silicon-gel chromatography (25% EtOAC/petroleum
ether) to afford the target compound (
Melting Point:
TLC: Rf (silica; ethyl
acetate: petroleum ether, 1:2) 0.55.
IR(KBr cm-1): 2996, 2944, 2841, 1694, 1637, 1580, 1462, 1417, 1230, 1020, 764.
1H-NMR (300 MHz, CDCl3): δ= 3.83 (s, 6H, (OCH3)2). 3.87 (s, 3H, OCH3). 4.00 (d, H,-CHCl-). 4.33 (s, H,-CH-). 4.55 (s, H,-CH-). 4.76 (d, H,-CH-). 6.38 (s,2H, PhH). 7.15~7.35 (m,5H,PhH).
Acknowledgment
The
author is grateful to Professor Xianming Hu of the State Key Laboratory of
Virology,
References
1. Hori, K.; Saito, S.; Kubota, K. B. J. Cancer 2002, 86 (10), 1604-1614.
2. Monk, K. A.; Siles, R.; Hadimani, M. B. Bioorg .Med. Chem. 2006, 14 (9), 3231-.
3. Neri, D.; Bicknell, R. Nat. Rev. Cancer 2005, 5 (6), 436.
4.
5. Hsieh, H. P.; Liou,
J. P.; Mahindroo, N. Curr. Pharm. Des. 2005, 11 (13), 1655.
6.
7. Tozer, G. M.; Prise, V.E.;
8. Banik,
I.; Becker, F. F.; Banik, B. K. J. Med. Chem. 2003, 46 (1), 12.
© 2006 MDPI. All rights reserved.