http://www.chemistrymag.org/cji/2001/037032ne.htm |
Jul. 1,
2001 Vol.3 No.7 P.32 Copyright |
Separation of a novel b-cyclodextrin derivative and its structure
Fang Zhijie(Chemical Engineering School, Nanjing University of Science & Technology, Nanjing, 210094 )
Recerived Mar.1, 2001; Supported by the National Natural Science Foundation of China (29972020).
Abstract A novel b-cyclodextrin(CD) derivative , heptakis(6I,II,III,IV,V,VI-O - tert - butyl dimethylsilyl)-b-cyclodextrin, was separated from the reaction mixture of CD with tert- butyl dimethylsilyl chloride(TBDMSCl) and the structure was suggested based on the data of 13C- NMR and elementary analysis.Keywords cyclodextrin derivative, separation, NMR
1 INTRODUCTION
Recently, it has been reported [1]a new synthetic strategy of
cyclooligosaccharides using cyclodextrins(CDs) as starting materials, and converting all
the glucopyranose units into another hexose units uniformly, might overcome the
difficulties in the syntheses and have a high possibility of shortest approach to the
aimed host molecules , because the availability of a variety of CDs will widen their use
as complexing agents and catalysts. Accordingly, we chose the synthetic route and studied
the synthesis of b-cycloatrins(CAs)
from b-CDs[2].
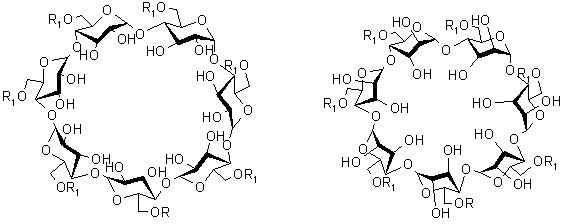 |
b-CD
b-CA 3 Scheme 1 |
In this paper, a novel b-CD (1) derivative, heptakis(6I,II,III,IV,V,VI- O - tert - butyl dimethylsilyl)-b-cyclodextrin(2 b) ,was separated from the mixture of heptakis(6- O - tert - butyl dimethylsilyl)-b-cyclodextrin(2a) and (2b) which formed when the b-CD reacted with tert – butyl dimethylsilyl chloride(TBDMSCl) forming compound (2a), the later as the intermediate to synthesize the final product b -CA (3), and the structure of (2a) and (2b) was suggested based on the data of 13C- NMR and elementary analysis.
2 EXPERIMENTAL AND RESULTS
2.1 General Methods
NMR spectra were recorded with Brucker WM 300 spectrometer, TLC(silica gel 60 F254
plastics sheets ,Merk) was used to monitor the reactions. Eluents employed: EtOAc : EtOH :
H2O (A = 50 : 2 : 1, B = 25 : 2: 1, C = 16 : 2 : 1, and D = 12 : 2 : 1) .The
spots were visualized with 50% sulfuric acid. Column chromatography was performed on
silica gel 60 (Merk 230-400 mesh).
2.2 Synthesis and separation of compounds 2 a and 2 b
Dry pyridine (30ml) was added to a flask by a
syringe. The weighed b-CD (2.28g, 2.01 mmol) was added to the stirred pyridine(4Å MS). A
solution of silyl chloride(2.38g, 15.81mmol) in dry pyridine(20ml) was then added to the b-CD solution dropwise at 0°C. The reaction mixture was stirred overnight at room temperature
and then poured into ice-cold water (200ml) and stirred vigorously for ca. 10 min. The
resulting precipitate was collected by filtration, washed with ice-cold water and dried
under vacuum to give 3.54g(94%) of the crude product. The crude product was purified by a
column chromatography using gradient elution with solvents A, B, C and D. Pure 2a (2.45g)
and a by-product 2b(0.31g) were obtained, yielding 63% and 8%, respectively.
2.2.1 Heptakis(6-O - tert- butyl
dimethylsilyl)-b-cyclodextrin(2a)
1H-NMR(CDCl3): δ = -0.01 (6H, s, SiCH3), 0.83 (9H, s, CH3),
3.53 (1H, t, J = 9.0 Hz, H-4), 3.60 (1H, dd, J = 3.4, 9.0 Hz, H-2), 3.60 (1H, d J = 9.6
Hz, H-5), 3.67 (1H, d, J = 10.8 Hz, H-6a), 3.86 (1H, dd, J = 2.4, 11.6 Hz, H-b), 4.00 (1H,
t, J = 9.0 Hz, H-3), 4.85 (1H, d, J = 3.4, H-1).
13C-NMR(CDCl3): δ = -4.60, -4.49 (Si-CH3), 18.86 (C-CH3),
26.49 (C-CH3), 62.21 (C-6), 73.13 (C-5), 73.99 (C-3), 74.19 (C-2), 82.35 (C-4),
102.59 (C-1).
Anal.calcd. for 2a (C12H24O5Si)7: C 52.14, H
8.75, Found: C 52.86, H 8.87, Lit.: C 51.54, H 8.52.
2.2.2 Heptakis(6I,II,III,IV,V,VI- O - tert – butyl dimethylsilyl)-b-cyclodextrin(
2b)
13C-NMR(CDCl3): δ =
101,87,102.10 ,102.27(C–1), 73,43(C-2), 72.66,73.30(C-3), 81.52, 81.72, 82.00,
82.67(C-4), 71.81, 72.25(C-5), 61.64, 61.98, 62.43(C-6).
Anal.calcd. for 2b (C78H154O35Si6): C
51.46, H 8.53, Found: C 52.08, H 8.45
3 DISCUSSION
The compound 2b has the further application possibility. In the branched
oligosaccharides, for example, this silyl-protected compound having only one free primary
OH group could be employed for synthesis of 6-a- D- glucopyranosylcycloheptaoside 2 c
as shown in the scheme 1, and it could be used to synthesize the promising compound as a
kind of sophisticated artificial receptors[3].
Acknowledgements
The author would like to express his sincere gratitude to Professor Dr. F. W.
Lichtenthaler for his continuous encouragement, Dr. E. Cuny for his helpful discussions.
REFERENCES
[1] Fujita K, Shimada H, Ohta K et al. Angew. Chem., 1995, 107: 1783.
[2] Fang Z J. Technical report, Technische Hochschule Darmstadt, 1996, 1-6.
[3] Venema F, Baselier C T, Feiters T C et al. Tetrahedrin Lett. 1994, 35: 8661.