http://www.chemistrymag.org/cji/2005/072016ne.htm |
Feb.1, 2005 Vol.7 No.2 P.16 Copyright |
Sun Wujuan, Yang Xuwu, Zhang Hongfang, Zhang
Hangguo, Li Na, Zhu Li, Gao Shengli
(Shaanxi Key Laboratory of Physico-Inorganic Chemistry, Department of Chemistry, Northwest
University, Xi'an Shaanxi 710069, China)
Received Nov. 12, 2004; Northwest University for financial support (No. 02NW02)
Abstract Rare earth(III)
complex with salicylic acid Eu(H2Sal)(HSal)(Sal)·H2O(H2Sal = C7H6O3,HSal = C7H5O3,Sal = C7H4O3)was synthesized, and it was
charaterized by IR and CV (Cyclic Voltammetry). The constant-volume combustion energy of
complex, ![]() , was determined as (-9029.50 ± 3.96 ) kJ·mol-1
by a precise rotating-bomb calorimeter at 298.15K. Its standard molar enthalpy of
combustion,
, was determined as (-9029.50 ± 3.96 ) kJ·mol-1
by a precise rotating-bomb calorimeter at 298.15K. Its standard molar enthalpy of
combustion, ![]() , and standard molar enthalpy of
formation,
, and standard molar enthalpy of
formation, ![]() , were calculated as (-9029.50 ± 3.96 ) kJ·mol-1
and (-2488.96 ± 43.82) kJ·mol-1 , respectively.
, were calculated as (-9029.50 ± 3.96 ) kJ·mol-1
and (-2488.96 ± 43.82) kJ·mol-1 , respectively.
Keywords rare earth, salicylic acid; cyclic voltammetry; constant-volume combustion energy; standard
molar enthalpy of formation
Nowadays great attention has been paid to the lanthanide aromatic carboxylates owing to its novel structures and potential applications in material sciences such as superconductor, magnetic materials, and luminescent probes [1-5]. Moreover, they are a kind of potential luminescent materials for further application. The luminescence, mechanism of thermal decomposition, stability constants and other properties of some lanthanide complexes with salicylic acid have been reported in literature [6-9]. However, thermochemistry of lanthanide complexes with salicylic acid has not been investigated. In this paper, the constant–volume combustion energy of the complex was determined by a precise rotating-bomb calorimeter at 298.15K, and its standard molar enthalpy of combustion and standard molar enthalpy of formation were calculated on the basis of the constant-volume combustion energy of complex. The final results would provide theoretical basis for enlarging their application fields.
1 EXPERIMENTAL
1.1 Reagents and apparatus
Europium chloride hydrate EuCl3·3.53H2O was prepared according to Ref. [10].
Salicylic acid and other reagents are of A. R. grade.
The C and H contents were measured by a Vario EL III CHNOS elemental
analyzer made in Germany; the rare earth ion was determined by EDTA volumetric analysis;
IR spectra was recorded with a Model EQUINOX55 FTIR spectrophometer ( KBr pellet); Cyclic
voltammograms were made with Potentiostat/Galvanostat Model 283 (EG&G company,
America), using with a three-electrode system with glass-carbon electrode as a working
electrode, SCE (saturated calomel electrode) as a reference electrode and a platinum wire
counter electrode; the constant-volume combustion energy of complex was carried out by a
RBC-type II precise rotating-bomb calorimeter; melting
point of the complex was measured with WRS-1A digital melting-point apparatus.
1.2 Preparation of complex
The complex was prepared according to Ref.
[11], 8.2873g salicylic acid was dissolved in NaOH solution with molar ratio of H2Sal:
NaOH=1: 1, and the salicylate sodium formed at 60℃.
6.4409g EuCl3·3.53H2O was dissolved in distilled water (the
molar ratio of EuCl3: NaHSal is 1:3), the solution of EuCl3·3.53H2O
was added dropwise into the solution of salicylate sodium at the condition of
electromagnetic stirring, pH was adjusted to 6.0, then the yellow precipitate was formed.
After cooling the solution, filtered, the solid product was washed by distilled water
repeatedly, then it was kept in a vacuum desiccator and dried by P4O10
until constant weight. The product was 10.4238g, and the yield was 89.7%.
2 RESULTS AND DISCUSSION
2.1 The analytical result of Eu(H2Sal)(HSal)(Sal)·H2O
Composition
Found (%): Eu 26.19, C 43.75, H 2.75;
Calc. (%): Eu 26.15, C 43.38, H 2.95.
2.2 IR spectra of the complex
The IR spectra of salicylic acid and the complex were
recorded over the range 4000-400cm-1. It can be seen from the IR spectra that
salicylic acid has five characteristic absorption peaks: nO-H (COOH, intramolecular hydrogen bonds, 3237.45cm-1),
nO-H
(COOH, intermolecular hydrogen bonds, 2857.57 cm-1), nO-H (COOH,
2596.33 cm-1), nC=O (COOH, 1660.76cm-1), dO-H (
phenolic hydroxyl, 1480.43 cm-1). IR spectra of complex has a hydroxyl
characteristic peak at 3289.31 cm-1, in which it appeared the symmetrical
stretching vibration absorption peak (nsCOO-,1387.32 cm-1)
and dissymmetrical stretching vibration absorption peak (nasCOO-,1560.93
cm-1) of carboxyl. It is assumed that
carboxyl disengaging the proton as acid radical coordinates to Eu3+. Three
modes which carboxyl coordinated with Eu3+ were obtained from the crystal
structure of the complex [12], some is monodentate coordination and others are
bidentate coordination (Fig.1), only parts of phenolic oxygen participate in coordination.
So there still are hydroxyl characteristic peaks in IR spectra of the complex, the
molecular formula for the complex is Eu(H2Sal)(HSal)(Sal)·H2O.
![]()
Fig.1 Three coordination modes of
salicylate with Eu3+
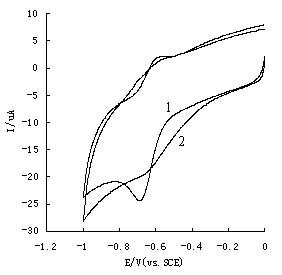
Fig.2 The cyclic voltammetry curves of Eu(H2Sal)(HSal)(Sal)·H2O
(curve 1) and EuCl3·3.53H2O(curve 2) on glass-carbon electrode
2.3 Electrochemical behavior
The test bottom liquid of electrochemistry was Tris-HCl buffer solution (pH=6.0). The
complex was dissolved with a small quantity of DMF and the solution was diluted to 1.6×10-4mol·L-1
with the bottom liquid. The oxygen was removed by aerating nitrogen 10 min. The scan scope
was 0.0 - -1.0V, the scan speed was 100mV/s. Then the cyclic voltammetry behavior of Eu(H2Sal)(HSal)(Sal)·H2O
was showed as Fig.2.
At potential scope of 0.0 - -1.0V (vs. SCE), the complex Eu(H2Sal)(HSal)(Sal)·H2O
(curve 1) has only a pair of redox peaks in the buffer solution of Tris-HCl (pH=6.0),
potential of reduction peak Epc=-0.698V, potential of oxidation peak Epa=-0.59V,
DE=108mV. This result is
similar to Eu(III)-tryptophane-benzimidazole ternary
complex[13], so it can be concluded that the electrochemical active center of
Eu(H2Sal)(HSal)(Sal)·H2O was Eu3+, and the electrochemical process
of the title complex was single electron redox process.
![]()
At the same time, the cyclic voltammetry
behavior of EuCl3·3.53H2O (curve 2) was also tested in the buffer solution
of Tris-HCl (pH=6.0). It also can be seen from the curve 2 that there was a pair of redox
peaks, the potential of reduction peak Epc=-0.658V, the potential of oxidation
peak Epa=-0.60V, DE=58mV. Moreover, we can find in the figure that the redox peaks of
dissociating Eu3+ was not very clear-cut, which indicates the electrochemical
reversibility of dissociating Eu3+ is stronger than the complex. It also can be
noticed that the reduction peak of complex is more negative than dissociating Eu3+,
which shows that the Eu3+ become steadier as the center of the complex, and the
electrochemical reversibility of complex is less than dissociating Eu3+.
2.4 Combustion energy of the complex
2.4.1 Experimental conditions
The constant-volume combustion energy of
the complex was determined by a precise rotating-bomb calorimeter (RBC-type II)[14].
The analytical methods and main experimental procedures were described previously[14].
The initial temperature was regulated to (25.0000 ±
0.0005) ℃, and the
initial oxygen pressure was 2.5 MPa. The correct value of the heat exchange was calculated
according to Linio-Pyfengdelel-Wsava formula[15]. The calorimeter was
calibrated with benzoic acid of 99.999 % purity. (Chengdu Chemical Reagent Company), which
has an isothermal heat of combustion of -26434 J·g-1 at 25℃, the calibrated experimental
results with an uncertainty 4.18×10-4 were (17775.09±7.43) J·K-1.
The analytical methods of final products (gas, liquid and solid) were the same as these in
Ref. [14], the analytical results of the final products showed that the
combustion reaction was complete. The calibrated experimental results were summarized in
Table 1.
Table 1 Result for Calibration of Energy Equivalent of the Rotating-bomb Calorimeter
No. |
Mass
of complex |
Calibrated heat of combustion wire qc/J |
Calibrated
heat of acid |
Calibrated |
Energy equivalent W/(J·K-1) |
1 |
0.99702 |
10.35 |
24.78 |
1.4834 |
17790.45 |
2 |
0.78940 |
8.10 |
20.89 |
1.1746 |
17789.88 |
3 |
0.83060 |
12.60 |
20.43 |
1.2382 |
17758.93 |
4 |
0.96869 |
12.60 |
17.43 |
1.4418 |
17780.82 |
5 |
0.99485 |
12.60 |
20.80 |
1.4800 |
17798.18 |
6 |
1.12328 |
9.09 |
21.85 |
1.6735 |
17761.41 |
7 |
0.90036 |
9.28 |
21.67 |
1.3429 |
17745.97 |
Mean |
17775.09±7.43 |
2.4.2 Constant–volume combustion energy of the complex
The methods of determination and calculation of the constant-volume combustion energy
for complex are the same as for the calibration of the calorimeter with benzoic acid. The
values are calculated by means of the following equation:
![]() (1)
(1)
where ![]() (complex, s) denotes the constant-volume combustion
energy of the complex (in J·g-1), W is the energy equivalent of
the rotating-bomb calorimeter (in J·K-1), a is the length of the
actual Ni-Cr wire consumed (in cm). G is the combustion enthalpy of Ni-Cr wire for
ignition (0.9 J·cm-1), 5.97 is the formation enthalpy and solution
enthalpy of acid corresponding to 1 mL of 0.1000 mol·L-1 solution of
NaOH (in J·mL-1), b is the volume (in mL) of consumed 0.1000 mol·L-1
solution of NaOH and D
(complex, s) denotes the constant-volume combustion
energy of the complex (in J·g-1), W is the energy equivalent of
the rotating-bomb calorimeter (in J·K-1), a is the length of the
actual Ni-Cr wire consumed (in cm). G is the combustion enthalpy of Ni-Cr wire for
ignition (0.9 J·cm-1), 5.97 is the formation enthalpy and solution
enthalpy of acid corresponding to 1 mL of 0.1000 mol·L-1 solution of
NaOH (in J·mL-1), b is the volume (in mL) of consumed 0.1000 mol·L-1
solution of NaOH and D
Table 2 Experimental Results for the Combustion Energy of the Complex
No. |
Mass
of sample |
Calibrated heat of combustion wire qc/J |
Calibrated heat of acid |
Calibrated |
Combustion
energy of sample |
1 |
1.14208 |
12.60 |
20.56 |
1.0003 |
15539.42 |
2 |
1.15003 |
12.60 |
20.70 |
1.0063 |
15524.62 |
3 |
1.14215 |
12.70 |
20.55 |
0.9988 |
15515.92 |
4 |
1.14380 |
12.60 |
20.59 |
1.0027 |
15553.32 |
5 |
1.13987 |
12.60 |
20.52 |
0.9966 |
15511.89 |
6 |
1.14522 |
12.60 |
20.61 |
1.0034 |
15544.89 |
Mean |
15531.68±6.81 |
2.4.3 Standard molar combustion enthalpy of
the complex
The standard molar combustion enthalpy of the complex, ![]() [Eu(C7H5O3)3
[Eu(C7H5O3)3
Eu(C7H5O3)3·H2O(s)+ 21O2(g)=
The standard molar combustion enthalpy of the complex is calculated by the following equations:
where ng is the total amount in mole of gases present as products or as reactants, R=8.314 J·K-1·mol-1, T=298.15K. The result of the calculation of
2.4.4 Standard molar enthalpy of formation for the complex
The standard molar enthalpy of formation of the compound,
where
The result of the calculation of
The standard molar enthalpy of formation of salicylic acid is -589.5kJ·mol-1 [17]. Compared with the enthalpy of formation for the complex, the latter is more negative; on the other hand, compared with its melting point, the melting point of the complex (>300℃) is higher than that of salicylic acid (157-159℃) [18], all these indicate that the complex is steadier than the ligand.
REFERENCES
[1] Costes J P,
Clemente-Juan J M, Dahan F et al. Chem. Int. Ed, 2002, 41: 323.
[2] Reineke T M, Eddaoudi M, Fehr M, et al. J. Am.
Chem. Soc, 1999, 121: 1651.
[3] Ma L, Evans O R, Foxman B M, et al. Inorg. Chem. 1999, 38: 5837.
[4] Seo J S, Whang D, Lee H, et al. Nature, 2000, 404: 982.
[5] Wang S, Pang Z, Smith K D L, et al. J. Chem. Soc., Dalton Trans., 1994: 955.
[6] Aoyama Y, Yamagishi A, Asagawa M. J. Am. Chem. Soc., 1988, 110: 4076.
[7] Tadashi M, Yadashi A. J. Chem. Soc. Chem. Commun., 1995: 1257.
[8] Brzyska W, Kula A. J. Therm. Anal, 1988, 34: 899.
[9] Turkel N, Ayolin R, Ozer U. Turk. J. Chem., 1999, 23 (3): 249.
[10] Su M Z, Li G P. Chemistry (Huaxue Tongbao), 1979, 43: 34.
[11] Li X S, Chen Y L, Liu J J. Chinese Rare Earths (Xi Tu), 2003 242: 71.
[12] Yin M C, Sun J T. Journal of Alloys and Compounds, 2004, 381: 50.
[13] Wu H X, Wang Z M, Wang H L, et al. Chinese Journal of Applied Chemistry (Yingyong
Huaxue), 1999, 16 (5): 37.
[14] Yang X W , Chen S P, Gao S L. J. Instrumentation Science & Technology, 2002, 30
(3): 311.
[15] Popov M M. Thermometry and Calorimetry. Moscow: Moscow University Publishing House,
1954: 382.
[16] Cordfunke E H P, Konings R J M. Therrnochimica Acta, 2001, 375: 65
[17] Dean J A. Lange's Handbook of Chemistry. 15th
Edi, Beijing: Beijing World Publishing Corporation/McGraw-Hill, 1999.
[18] China Medicine (Group) Shanghai Chemical Reagent Corporation. The Handbook of
Reagents. 2nd Edi, Shanghai: Shanghai Technology Press, 1985: 1076.
配合物Eu(H2Sal)(HSal)(Sal)·H2O的热化学性质研究
(西北大学 化学系/陕西省物理无机化学重点实验室, 陕西 西安 710069)
摘要 合成了稀土水杨酸配合物Eu(H2Sal)(HSal)(Sal)·H2O(H2Sal= C7H6O3,HSal=C7H5O3,Sal=C7H4O3),通过红外,循环伏安法对配合物进行表征。在298.15K下,用精密转动弹热量计测定出配合物的恒容燃烧能
关键词 稀土,水杨酸,循环伏安法,恒容燃烧能,标准摩尔生成焓