http://www.chemistrymag.org/cji/2006/086044ne.htm |
Jun. 8, 2006 Vol.8 No.6 P.44 Copyright |
AM1 Investigation on Stabilities of C26BN Isomers
Li Jianhong, Wang Zhichang, Zhang Xie, Li Jianqiang(Department of Applied Chemistry, East China Institute of Technology, Fuzhou, 344000)
Abstract The possible stable
structures of substituted fullerene C26BN formed on the initial C28
cage with Td symmetry have been systematically studied by AM1 method included
in Gaussian 98W package. The symmetries(Cs or C1) were constrained
and the charge and multiplicity of all the isomers are zero and five, respectively. The
geometrical optimization and the vibrational frequencies analysis were performed on the
same level with constrained symmetry(Cs or C1). The results show
that the most stable isomer of C26BN is formed by boron and nitrogen atoms
doping at 5,2-sites. The stabilities of C26BN isomers were determined mainly by
B-N and conjugative effect.
Keywords C26BN isomers; AM1; stability
1 INTRODUCTIOM
Heterofullerenes which have one or more carbon atoms substituted by other elements
such as boron and nitrogen are expected to be used as semiconductors and building
materials for nanometer electronics due to their potential novel properties [1-2],
such as superconductivity, hardness, photoinduced electron transfer and nonlinear. The
first heterofullerene of C60-xBx(x=1-6) were prepared by Smalley[3]
and co-worker in 1991.
Considering BN and CC are isoelectronic species, boron and nitrogen
atoms substituted fullerenes may be stable, which were also surmised from a large number
of research results. In fact, BN substituted C60-n(BN)n have already
been reported by Piechota early in 1996. Theoretical studies on the structures and
stabilities of C34BN isomers[4], C38BN[5] have
been performed recently. AM1 method is proved to be an effective means to forecast the
structure and stabilities of the heterofullerene isomers[4]. However, it turns
out that little have been known so far about the boron and nitrogen substituted C26BN
based on fullerene C28 with Td symmetry[5] and four
unpaired electronics on ground state, the smaller fullerene relatively to C36
and C60.
2 COMPUTATIONAL METHODS
The C26BN isomers were constructed by boron and nitrogen substituted the
carbon atoms of the C28 cage with Td symmetry. The geometrical
optimization and the frequencies of all the possible isomers of C26BN have been
performed at the AM1 level supplied by Gaussian 98W package on P4 computer, the keyword
scf(conver=9) was used in the calculations, the charge and the multiplicity of the isomers
of C26BN are zero and five, respectively. Symmetry (Cs or C1)
of all the isomers were constrained during the optimizing process.
3 RESULTS AND DISCUSSION
The geometry configuration of C28 with Td symmetry was shown in
Fig.1. For the convenience of discussion, all the carbon atoms of C28 cage were
numbered, and heterofullerenes C26BN were labeled as C26BN: i-j
which indicated that boron the special symmetry, C28 molecule with Td
symmetry has three kinds of atoms, the first kind of carbon atoms labeled as 1,8,15 and 22
are the juncture of three five-membered rings, the second kind of carbon atoms connect the
first kind of carbon atoms labeled as 2,3,4, and so on, the third kind connect the second
kind. Except the isomers with the same energy brought by symmetry, forty C26BN
isomers were obtained. The symmetry and heat of formation(HOF) at AM1 level were listed in
Table 1. The vibrational analysis indicated all the isomers have no imaginative
frequencies.
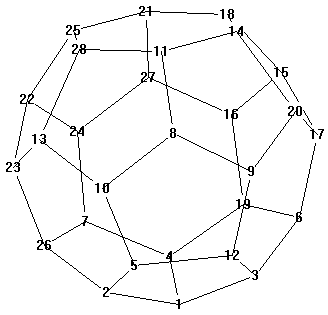
Figure 1 The structure and numbering of C28
with Td symmetry
Table 1 The symmetry and heat of formation for C26BN isomers at AM1 level
No. |
Isomers |
Sym. |
HOF |
No. |
Isomers |
Sym. |
HOF |
1 |
C26BN: 1-2 |
Cs |
3399.546 |
21 |
C26BN: 11-2 |
C1 |
3506.737 |
2 |
C26BN: 1-5 |
C1 |
3452.388 |
22 |
C26BN: 13-2 |
Cs |
3418.987 |
3 |
C26BN: 2-5 |
C1 |
3410.013 |
23 |
C26BN: 14-2 |
C1 |
3415.022 |
4 |
C26BN: 2-6 |
C1 |
3476.896 |
24 |
C26BN: 14-3 |
Cs |
3412.698 |
5 |
C26BN: 2-7 |
C1 |
3489.323 |
25 |
C26BN: 8-4 |
Cs |
3451.153 |
6 |
C26BN: 2-1 |
Cs |
3425.170 |
26 |
C26BN: 9-4 |
C1 |
3439.566 |
7 |
C26BN: 5-1 |
C1 |
3442.078 |
27 |
C26BN: 11-4 |
Cs |
3443.259 |
8 |
C26BN: 2-3 |
C1 |
3534.340 |
28 |
C26BN: 13-4 |
C1 |
3414.304 |
9 |
C26BN: 5-2 |
C1 |
3363.465 |
29 |
C26BN: 12-5 |
Cs |
3487.880 |
10 |
C26BN: 6-2 |
C1 |
3414.490 |
30 |
C26BN: 13-5 |
C1 |
3451.747 |
11 |
C26BN: 7-2 |
C1 |
3426.489 |
31 |
C26BN: 14-5 |
C1 |
3440.147 |
12 |
C26BN: 5-6 |
C1 |
3439.850 |
32 |
*C26BN: 8-6 |
C1 |
3441.286 |
13 |
C26BN: 8-1 |
Cs |
3541.287 |
33 |
C26BN: 9-6 |
Cs |
3480.335 |
14 |
C26BN: 10-1 |
C1 |
3467.902 |
34 |
C26BN:10-6 |
C1 |
3481.347 |
15 |
C26BN: 11-1 |
Cs |
3471.044 |
35 |
C26BN: 13-6 |
C1 |
3484.843 |
16 |
C26BN: 13-1 |
Cs |
3438.183 |
36 |
C26BN: 14-6 |
C1 |
3480.197 |
17 |
C26BN: 14-1 |
Cs |
3462.749 |
37 |
*C26BN: 8-7 |
C1 |
3520.526 |
18 |
C26BN: 8-2 |
C1 |
3449.878 |
38 |
C26BN: 9-7 |
C1 |
3488.314 |
19 |
C26BN: 9-2 |
C1 |
3497.104 |
39 |
C26BN: 10-7 |
Cs |
3488.535 |
20 |
C26BN: 10-2 |
C1 |
3453.640 |
40 |
C26BN: 13-7 |
C1 |
3439.850 |
As far as C26BN isomers are concerned, heteroatoms bring about deformation more or less, the deformation of C26BN isomers with bonded heteroatoms is small, whereas the deformation of C26BN isomers with isolated heteroatoms is large. The C26BN: 5-2 isomer has very similar structure to C28(Td) and is the most stable isomer. The difference of boron and nitrogen atomic charge is affected mainly by the distance of boron and nitrogen atoms, when boron and nitrogen are bonded or near, s-bond inducing effect makes electron transfers from boron atom to nitrogen atom, this is primary; on the other hand, conjugative effect makes electron transfers from carbon and nitrogen atoms to boron atom, because two electrons of nitrogen atom, one electron of carbon atom and empty orbit supported by boron atom participate in the conjugative effect. It can be concluded that the stabilities of the C26BN isomers are affected by both inducing effect and conjugative effect, since the bonded boron and nitrogen atoms come up to one carbon-carbon bond to a great extent. This shows that bonded B-N unit make a great contribution to the stabilities of the C26BN isomers, which is in good agreement with the former conclusion[7]. When nitrogen atom and boron atom replace C2 and C5 atom, respectively, nitrogen atom can provide electron both to boron and also to C1 atom, this forms remarkable conjugative effect, keeps the analogous structure of C28(Td), whereas, boron atom of another isomer C26BN: 2-5 cannot provide electron to C1 atom to conjugate although nitrogen can provide electron to boron atom. The heat of formation of the other isomers with unbonded heteroatoms are high, greater than the most stable one due to lack of B-N σ-bond inducing effect and lack of conjugative effect. What is more, It is worthwhile to mention that C26BN: 12-5 isomer has a considerably high heat of formation although boron and nitrogen atoms are adjacent, yet this substitution mode will destruct the conjugative effect.
REFERENCES
[1] Satoshi Nakano, Minoru Akaishi, Takayoshi Sasaki et al. Chem. Mater., 1994, 6:
2246-2251.
[2] Yao B, Liu B, Su WH. J. Mater. Res., 1998, 13(7): 1753-1756.
[3] Guo Ting, Jin Changming, and R. E. Smalley, Doping bucky: formation and properties of
boron-doped buckminsterfullerene[J]. J. Phys. Chem., 1991, 95(13): 4948-4950.
[4] Yang Xia, Wang Guichang, Shang Zhenfeng et al. Phys. Chem. Chem.. Phys.,
2002,4:2546-2553.
[5] Lu Li-HWA, Sun Kuang-Chung, Chen Cheng. Int. J. Quant. Chem., 1998, 67(3):
187-197.
李建红, 王志畅, 张燮, 李建强
东华理工学院应用化学系, 抚州344000
摘要 用半经验的AM1方法对C28(Td)的等电子体C26BN所有可能的构型进行了优化,在优化过程中保持了异构体的对称性(Cs or C1),所有异构体的电荷和多重度分别是0和5,同时进行了振动频率分析。结果表明最稳定的异构体是C26BN: 5-2。C26BN各异构体的稳定性主要由体系的B-N键诱导效应和共轭性质决定。
关键词 C26BN异构体;AM1;稳定性