http://www.chemistrymag.org/cji/2001/03b056ne.htm |
Nov. 1,
2001 Vol.3 No.11 P.56 Copyright |
Tan Shengjian, Peng Qingtao, Hu Wenxiang,*
Chen Peirang
(Institute of Military Medicine, Headquarters of General Equipment, Beijing 100101)
Received July.1, 2001
Abstract A RP-HPLC method for the
determination of imperatorin in Yuanhu analgesic capsule was established. The sample was
dissolved in ethanol and extracted by refluxing in water bath , then chromatographed on
LUNA-C18 column with mobile phase of methanol-0.5% phosphoric acid (69:31) .
The flow rate was set at 0.8mL/min and the detection wavelength was of 300nm. The
resolution between imperatorin and its neighboring peak was 2.0, the number of theoretical
plates was 15367 according to the peak of imperatorin. The linear range was 19.50-58.50mg/mL, r was 0.9999, and the average recovery was 99.8%. This HPLC
method is rapid and accurate, it can be used to determine imperatorin quantitatively in
Yuanhu analgesic capsule.
Keywords RP-HPLC, Yuanhu analgesic capsule, Imperatorin
Yuanhu analgesic capsule is made of Yuanhu and Radix Angelicae Dahuricae which is recorded in the eighth volume (1993) of Medicine Standards published by the Ministry of Health of the People's Republic of China. Its function is to regulate the flow of vital energy and remove obstruction to it, to invigorate the circulation of blood and to relieve pain. It can be used to treat stomach pain caused by stagnation of the circulation of vital energy and blood stasis, to treat rib pain, headache and menses pain and so on. Imperatorin is the main active component of Radix Angelicae Dahuricae [1]. Quality of Yuanhu analgesic capsule is identified by the characteristics and general rule of capsule examination in Medicine Standards. It was reported[2,3] that the concentration of imperatorin in Radix Angelicae Dahuricae of some medicines was determined by HPLC, but the determination method of imperatorin in Yuanhu analgesic capsule has not been reported. In this study, a RP-HPLC method for the determination of imperatorin in Yuanhu analgesic capsule was established.
1 INSTRUMENT AND REAGENTS
The HPLC system consists of LC-6A liquid pump of Shimadzu Corp. of Japan, SIL-6B automatic
sample injector, SPD-6AV ultraviolet-visible spectrophotometer detector and Chromatopac
C-R 6A data processor. Medicine for experiment was purchased from Medicine Corp. of
Beijing and was identified by us. The standard sample of imperatorin was purchased from
the Medicine and Biological Products Identification Institute of China. Yuanhu analgesic
capsules were made by the Medicine Identification Institute of the Second Artillery of PLA
. Methanol was of HPLC grade and ethanol was analytical reagent. The stock solution of
imperatorin standard sample was ethanol solution of 97.50m g/mL.
2 METHOD AND RESULTS
2.1 Chromatographic conditions
LUNA C18 column (4.6mm ID×250mm, particle
diameter 5m m), ODS protective column(5mm ID×50mm, particle
diameter 5mm) of Analytical Apparatus Factory of Beijing were used . The column
temperature was at 35°C. The mobile phase was the mixture of methanol-0.5% phosphoric
acid (69:31) and the flow rate was set at 0.8mL/min. The detection wavelength was of
300nm, the injected volume was 10mL and the recorder paper rate was 1mm/min.
2.2 Experiment operations
The standard sample of imperatorin was diluted to suitable concentration by ethanol, then
injected. The negative sample without imperatorin was prepared according to Yuanhu
analgesic capsule prescription. The sample of Yuanhu analgesic capsules and the negative
sample were taken and extracted according to the sample determination method, injected and
the chromatograms were given in Figure 1. From the chromatograms, it was known that the
other components of Yuanhu analgesic capsule did not affected the determination of
imperatorin. The retention time of imperatorin was about 16 min, the resolution between
imperatorin and its neighboring peak was 2.0, the number of theoretical plates was 15367
according to the peak of imperatorin. Under the chromatographic conditions to be examined,
imperatorin in Yuanhu analgesic capsule can be determined quantitatively.
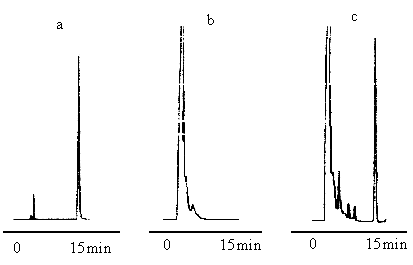
Fig.1 The chromatogram of (a) imperatorin
(b) negative sample of Yuanhu analgesic capsule (c) Yuanhu analgesic capsule, the
peak of 1 in chromatogram (c) is imperatorin
2.3 Linear relation and the minimum detect
limit
The standard sample was taken precisely and diluted to different concentration(19.50~58.50mg/mL) with ethanol, then injected 10mL respectively and the peak area was recorded. The linear
regression was made between the mean peak area of imperatorin X and the injected
concentration Y(mg/mL). The
regression equation:
Y=0.000070964X+3.1591; r=0.9999.
The minimum detect limit was 0.1mg/mL according to the three times noise of baseline.
2.4 Extraction method
The extraction rates of imperatorin by ethanol were examined by using different ultrasound
time and different reflux time in water bath. Our results shown that the extraction rate
was 96.3% by ultrasounding for 60min and that of by refluxing in water bath for 20min was
100%. Therefore, the extraction method of refluxing in water bath was selected. If take
the extraction rate as 100% by refluxing for 40min, then the extraction rate was 99.9%,
100.5% and 98.3% respectively by refluxing for 20, 60, 90min.
2.5 Apparatus precision and method repeatability
A sample solution was taken into the automatic sample injector, and injections were made
in 5h for n=5, interday RSD% was 0.3170. The same sample was determined for 3 consecutive
days and five times per day (n=5), intraday RSD% was 3.864.
2.6 Adding standard recovery
Six parts of the same batch Yuanhu analgesic capsules were weighed precisely, among them,
imperatorin standard sample was added precisely into five parts, the other part was the
blank. Every part was operated according to the sample determination method for
determining the imperatorin concentration. The average recovery was 99.8%, RSD% was 0.63
(n=5).
2.7 Sample determination
Fifteen Yuanhu analgesic capsules were taken and ground into powder. A certain amount of
the powder was precisely weighed (about equivalent to Radix Angelicae Dahuricae 0.9g) into
a 100mL Erlenmeyer flask with ground cap and 50mL ethanol was added. The mixture was
extracted by refluxing for 40min in water bath, then the mixture was cooled down to room
temperature and was filtered. 25mL continuous filtrate was precisely measured into a 50mL
volumetric bottle, ethanol was added to the scale and the bottle was shaked evenly. A
little was taken from the volumetric bottle and filtered through a 0.45mm membrane. An aliquot of 10mL was injected into the HPLC, the peak
of imperatorin was recorded and its amount was calculated by the external standard method.
Three batches samples were determined, the results of imperatorin was 0.66, 0.71 and
0.72mg/capsule respectively.
3 DISCUSSION
By the stability experiment, it can be concluded that the concentration of imperatorin in
the sample solution was stable at least for 48h, which was placed in brown volumetric
bottle at 18°C.
Different proportions of mobile phase of methanol-water and absorption
wavelength were experimented repeatedly, the result showed that it could not obtain
baseline separation for imperatorin with other components in 25 min until methanol-0.5%
phosphoric acid (69:31) was selected as mobile phase.
REFERENCES
[1] Yin Jian, Guo Ligong. Modern Research Medica and Clinical Application of Chinese
Materia, Volume 1, Beijing: Xue Yuan Publisher, 1995: 259.
[2] Chen Yianxiang, Shen Anhua. Research of Chinese Traditional Patent Medicine, 1991, 11
(6): 26.
[3] Huang Xinsheng. Chinese Traditional and Herbal Drugs, 1988, 6: 19.