http://www.chemistrymag.org/cji/2003/059071ne.htm |
Sep. 1, 2003 Vol.5 No.9 P.71 Copyright |
Microwave-assisted acetalization of pentaerythritol catalyzed by ammonium hydrogen sulfate
Zhang Jinmei, Li Yiqun(Department of Chemistry, Jinan University, Guangzhou 510632)
Received Jul. 14, 2003. Abstract Efficient conversion of carbonyl compounds, aldehyde or ketone, with pentaerythritol to the corresponding compounds in the presence of ammonium hydrogen sulfate under microwave irradiation without solvent is described.
Keywords diacetal of pentaerythritol, microwavae irradiation, ammonium hydrogen sulfate.
The diacetal of pentaerythritol are a series of useful organic compounds. They have been used as plasticizers and vulcanizer, as active physiologically substance, and as potential protective groups in organic synthesis. The preparation of these compounds has been well investigated. A variety of acidic catalysts, such as hydrochloric acid, sulfuric acid, p-toluenesulfonic acid, zinc chloride, acidic cation exchanger, Montmorillonite clay
[1], expansive graphite[2]and anhydrous ferrous sulfate[3] have been used for this purpose.Microwave dielectric heating has recently attracted much attention and become an important technique in organic synthesis[4]. Recently p- toluenesulfonic acid[5] in dry media, 12-tungstophosphoric acid[6] in acetonitrile coupled with microwave irradiation has been proven to be an efficient method for the synthesis of diacetal of pentaerythritol. However these methods are not entirely satisfactory, due to such drawbacks as using organic solvent, corrosive acid, or tedious work-up, etc. Therefore, there is a need for developing an efficient and convenient catalytic method for the reaction using inexpensive and non-polluting reagent.
Surprisingly the potentiality of ammonium hydrogen sulfate as a catalyst has not, hitherto, been studied in the synthesis of diacetal of pentaerythritol. We have found that this common commercial available reagent can catalyzed the reaction under the microwave irradiation to produce the corresponding diacetates in good purity and moderate yields. The results are summarized in the Table 1,Table 2 and Table 3. Scheme 1
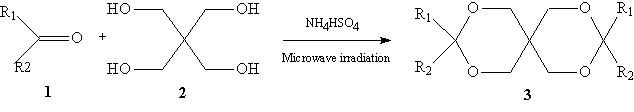
Table 1 Microwave-assisted acetalization of pentaerythritol
Entry |
Aldehydes |
Power |
Time (min) |
m.p.b |
Lit.m.p. |
Yields |
a |
n-C3H7CHO |
750 |
1.2 |
44-45 |
43-44[7] |
63 |
b |
C6H5CHO |
750 |
1.5 |
157-158 |
159-160[6] |
72 |
c |
4-Cl-C6H5CHO |
750 |
1.5 |
197-199 |
199-200[6] |
60 |
d |
cyclohexanone |
750 |
1.2 |
114-115 |
115-116[6] |
64 |
e |
2-NO2-C6H5CHO |
750 |
2.4 |
165-167 |
163-164[5] |
56 |
f |
4-MeO-C6H5 |
750 |
1.0 |
184-185 |
180-182[3] |
43 |
g |
2-Furylaldhyde |
750 |
0.6 |
162-163 |
158-159[6] |
72 |
h |
3-NO2-C6H5CHO |
750 |
2.0 |
185-186 |
185-186[3] |
63 |
Table 2 IR and NMR spectral data
Compd. |
IR (KBr,cm-1) |
1 H-NMR (CD3COCD3, d) |
3a |
2963,2865,1464,1394,1161,1099,1029 |
0.89(6H,t),1.39(4H,m),1.51(4H,m), |
3b |
3036,2995,2919,2854,1457,1386,1078,1028 |
3.50-3.95(6H,m), 4.80(2H,d), 5.42(2H,s), |
3c |
748,702,2921,2861,1383,1077,1025,814 |
3.20-3.60(6H,m),4.31(2H,s),4.55(2H,d), |
3d |
2936,2859,1445,1370,1251,1160,1087,1043,917 |
1.20-2.0(20H,m), 3.20-3.60(6H,m), |
3e |
2969,2917,2858,1526,1356,1070,737 |
3.21-3.60(6H,m),4.32(2H,s),4.56(2H,d), |
3f |
3010,2965,2851,1614,1517,1456,1388, |
3.20-3.60(6H,m)3.92(6H,s),4.30(2H,s), |
3g |
3126,2979,2921,2864,1504,1460, |
3.50-3.95(6H,m), 4.80(2H,d), 5.55(2H,s) |
3h |
3082,2978,2857,1620,1526,1347,1080,732,682 |
3.21-3.62(6H,m),4.32(2H,s),4.56(2H,d),6.80-7.90 (6H,m) |
Power (W) |
Time (min) |
Yields (%)a |
750 |
1 |
60 |
750 |
1.5 |
72 |
750 |
2 |
62 |
637 |
2 |
68 |
495 |
2 |
52 |
In conclusion, we provide an alternative protocol for the preparation of diacetals from aldehydes, because of its simplicity in operation, moderate yields, short reaction time and minimal environment impact.
EXPERIMENTAL SECTION
Melting point are uncorrected and determined on WRS-1 digital melting point apparatus
in open capillary. IR spectra were recorded using KBr pellet on Bruke Equinox 55
sectrometer. 1H NMR spectra were recorded in acetone on Unity Inova 500 (500
MHz) with TMS as an internal standard. Benzaldehyde and Furyl aldehyde was purified by
distillation. All other chemicals used were of commercial grade without further
purification.
Typical procedure: A mixture of carbonyl compound (10 mmol),
pentaerythritol (5 mmol) and sulfamic acid (2 mmol) was introduced into a Galanz WP 750A
domestic microwave oven in an open 25ml vessel. Microwave irradiation was carried out for
0.6-2.4 minutes at 495-750W power, followed by adding a lot of cool water and shaking the
vessel rigorously to precipitate the solid. The solid materials was filtered off and
treated with saturated sodium hydrogen sulfate solution, and then washed with water for
several times until the filtrate became neutral. The crude products were purified by
recrystallization to give the title compound.
REFERENCES
[1] Zhang Z H, Li T S, Jin T S et al. J. Chem.Res.(S), 1998, 640-641.
[2] Jin T S, Li T S, Zhang Z H. Synth.Commun.1999, 29: 1601-1606.
[3] Jin T S, Ma Y R,Li T S et al. J. Chem. Res.(S), 1999, 268-269.
[4] Varma, R. S.Solvent-free organic syntheses. Green Chem., 1999, 1: 43-55.
[5] Wang C D, Shi X Z, Xie R J. Synth. Commun., 1997, 27: 2517-2520.
[6] Peng Y Q, Song G H, Qian X H. Synth.Commun., 2001, 31: 3735-3738.
[7] Chen X,Xu Y T,Jin C X. Synth. Chem. (Hecheng Huaxue), 1997, 5 (2): 212-214.