http://www.chemistrymag.org/cji/2005/07b076ne.htm |
Nov. 25, 2005 Vol.7 No.11 P.76 Copyright |
(Research Center of Physics and Chemistry Analysis, Hebei University; Hebei Province Key Laboratory of Analytical Science and Technology, Baoding 071002 China) Received on Aug. 29, 2005. Abstract The separation and simultaneous quantitative determination of salicylic acid, phenol and resorcinol in Compound Fluocinnonide Tincture using reversed-phase ion-pair high-performance liquid chromatography on a C18(150*4.6mm i.d.,5mm)column was investigated. A mixture of methanol-acetonitrile-water10:25:65(v/v/v) containing 1.0mmol/L tetrabutyl ammonium bromide was used as the mobile phase and the detection wavelength was 225nm.The average spiked recoveries were all above 85.0% and the relative standard deviations were between 0.48% and 3.34%.The calibration curve was linear between0.014mg/ml to 5.40mg/mL, and the linear relativity were 0.9971, 0.9997 and 0.9977. The limits of detection were 0.014mg/ml,0.027mg/ml,0.019mg/ml, respectively.
Keywords Compound Fluocinonide Tincture; salicylic acid; phenol ; resorcinol; reversed-phase ion-pair chromatography The Compound Fluocinonide Tincture can kill germs and diminish inflammation. It is widely used for the treatment of neurodermatitis and is also effective in curing psoriasis in a sense.It consists of many components, such as fluocinonide, salicylic acid, resorcinol, borneolum syntheticun, ethanol et al .During the course of production and transportation, salicylic acid would probably be degradable to phenol with the existence of violet[1],and resorcinol is easily oxidated , which make the color of the Tincture solution deeper and deeper, and the effect of the Tincture decreased. Establishing an effective and quick method to monitor the quality of the Tincture is of great importance. Currently a number of methods have been described for the determination of salicylic acid or resorcinol, which include neutralization titration[2], ultraviolet spectrophotometry ,HPLC[3-5],capillary zone electrophoresis[6] ,gas chromatography [7]et al. But some of these methods could not simultaneously detect them. The goal of this article was to develop an alternative method for the separation and simultaneous determination of salicylic acid, phenol and resorcinol. The method here was proved simple, direct, valid and sensitive for the detection of these three phenolic compounds and the results were satisfactory. 1. EXPERIMENTAL
1.1 Apparatus
The HPLC system consisted of LC-10AT solvent delivery pump, 7725i injector and SPD-10A UV-VIS detector from Shimadzu Corporation(Japan).The N-2000 Double-Channel Chromatograph Data Operation Station and Software were from ZheJiang University Intelligent Information Engineering Research Institute. The UV-265 spectrophotometer was from Shimadzu Corporation (Japan).
1.2 Reagents and standards
Salicylic acid, phenol, resorcinol and tetrabutyl ammonium bromide were of analytical reagent grade and were used without further purification. HPLC grade solvents such as methanol and acetonitrile were used to prepare the mobile phase. Compound Fluocinonide Tincture was from a Shanghai Pharmaceutical Company,with the batch number 050401.Water used was distilled twice by passing through SZ-93 pure water system.
Stock solutions were fresh weekly prepared individually by dissolving 156.5mg Salicylic acid, 149.1mg phenol and 64.7mg resorcinol with the mobile phase in 100ml volumetric flasks. All stock solutions were stored at 4oC in a refrigerator and would be diluted to the required concentrations before being injected directly into the chromatograph.
1.3 Chromatographic conditions
Separation was performed using Kromasil C18(150*4.6mm i.d., 5μm)(Nobel Eka ,Sweden).The temperature of the column was kept at room temperature. The mobile phase consisted of methanol-acetonitrile-water10:25:65 (v/v/v) containing 1.0mmol/l tetrabutyl ammonium bromide. The detection was carried out at the wavelength of 225 nm.The flow rate was 1.0ml/min and the injection volume was 5.0ml.
1.4 Sample preparation
The Compound Fluocinonide Tincture (with the batch number 050401) was transferred accurately 2.0ml with a pipet. It was extracted with 1.0 ml petroleum ether, and the supernatant was discarded. Then the extracted layer was extracted again with 1.0 ml petroleum ether. Again the supernatant was discarded and 1.0ml distilled water was added to the extracted layer. The solution stratified, and the supernatant was discarded. The extracted layer was filtered with a syringe filter, diluted to the final concentration with mobile phase and injected into the chromatography system.
2. RESULTS AND DISCUSSION
2.1 Detection wavelength
The UV spectra of these three phenolic compounds were recorded from 200nm to 400 nm, and the wavelengths corresponding to UV maximums of these standard compounds were: salicylic acid (225 nm), phenol (265 nm and 221 nm),resorcinol (278 nm and 227.5nm) respectively. In order to achieve a better sensitivity and less interference, the detection was performed at 225nm in sensitivity range 0.01 AUFS.
2.2 Selection of mobile phase
To obtain satisfactory resolution and to avoid peak tailing of the compounds , an optimization of the proposed method was carried out using different mobile phase. Mobile phases of various compositions of methanol and water were tested. Using the system of methanol-water 20:70 and 40:60 (v/v ) the peaks of resorcinol and phenol were not well separated, and the retention time of resorcinol was too short. With an increase of the volume of acetonitrile added into the mobile phase, all the compounds could be separated but the retention time of resorcinol was still too short. Since these phenolic compounds were all weak acids and could ionize to a certain extent, using the ion-pair chromatography could improve the retention and separation. Different concentration of tetrabutyl ammonium bromide were added into the mobile phase, and the effect on elution behavior was investigated. The results indicated that with the increasing concentration of the tetrabutyl ammonium bromide, the retention time delayed accordingly. But when the concentration was 2.0 mmol/l or more , the retention time of resorcinol was shortened. The best results were obtained using the mobile phase methanol-acetonitrile-water 10:25:65(v/v/v) containing 1.0mmol/L tetrabutyl ammonium bromide, and the analysis could be finished within 8 minutes. The chromatogram of the standard compounds is shown in Fig 1.
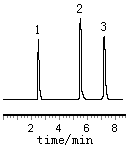 |
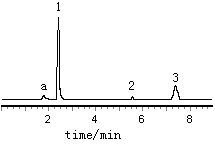 |
Fig 1
Chromatogram of standard compounds |
Fig 2
Chromatogram of the sample |
2.3 System suitability
System suitability tests were performed and chromatographic parameters were calculated
from experimental data, such as the number of theoretical plate and the resolution. The
number of theoretical plate for resorcinol was 2500 and the resolution was 3.3 between
resorcinol and the unknown compound according to the chromatogram of the sample.
2.4 Optimization of the sample preparation
Some components such as fluocinonide and borneolum syntheticum in the sample also had
ultraviolet absorption at 225nm and it interfered with the detection of the phenolic
compounds .To eliminate the interferences in the detection, the sample preparation was
optimized. The sample solution was initially only extracted with 2.0ml petroleum ether and
filtered with a syringe filter before detection . It was observed that although the sample
preparation step was simple, some components that interfered with the detection of the
phenolic compounds could not be eliminated and the reliability of the method could not be
checked. When 1.0ml distilled water was added into the solution that purified by petroleum
ether, the solution stratified evidently. The supernatant was discarded and the extracted
layer filtered. Before injected into the chromatographic system, the solutions were
diluted with mobile phase. Under the optimal chromatographic conditions, the components
that interfered with the detection were eliminated, and the concentrations of salicylic
acid, phenol and resorcinol were detected. The mechanism is likely that these phenolic
compounds are water-soluble compounds but other major components in the sample can only be
dissolved in water with the presence of other organic solvents and the addition of water
broke their capacity of dissolving in water. Thus the interferences of fluocinonide ,
borneolum syntheticum and other components in the sample solution could be eliminated.
2.5 Linear equations, correlation coefficients and detection limits
A series of working solutions were prepared by
diluting the stock solutions with mobile phase to the final concentrations and they were
detected under the optimized chromatographic conditions. The calibration graphs of the
standard compounds were found to be linear over the concentration range studied. The
linear equations and the detection limits of the three compounds are listed in Table 1.
(Y-peak area; X-the concentration of the detected compound)
Table 1 Linearity of the determination of three phenolic compounds
Components |
Linear equations |
Correlation |
Linear ranges |
Detection limits |
Salicylic acid |
Y=976.15+5930.42X |
0.9971 |
0.019-3.90 |
0.019 |
2.6 Recoveries and precision
The recoveries of the method were studied .
The stock solutions of different concentrations were added in the Compound Fluocinonide
Tincture solution . Then the sample solutions were prepared with the same method as
described in 1.4 and diluted with mobile phase before detection. The
concentrations of the phenolic compounds were examined and the recoveries could be
calculated according to the linear equation. The intra-day precision of the method was
determined by preparing the sample with the same method as described in 1.4 for each
analysis and the values were detected by 5 repeated analysis in a single day. The sample
solutions were fresh daily prepared for the determination of the inter-day precision of
the method which was checked by determining each phenolic compound during 5 days. The
results are given in Table 2.
2.7 The determination of the sample
The applicability of the method for the simultaneous determination of salicylic acid,
phenol and resorcinol was verified by the determination of them in the Compound
Fluocinonide Tincture. The sample solution was prepared with the same method as described
in 1.4. Then the sample solution was diluted 40,000 times, 40
times, 200,000 times respectively with mobile phase before detection and the
concentrations of salicylic acid, phenol and resorcinol were determined by 5 repeated
analysis. The results are given in Table 3. The chromatogram of the sample is shown in Fig
2.
Table 2 Recoveries and assay precision
Components |
Added contents |
Determined contents |
Recoveries /% |
Mean /% |
Intra-day |
Inter-day |
Salicylic acid |
1.25 |
1.14 |
91.29 |
|||
1.57 |
1.45 |
92.25 |
92.09 |
3.34 |
2.76 |
|
1.88 |
1.74 |
92.75 |
||||
Phenol |
1.49 |
1.39 |
93.57 |
|||
1.86 |
1.73 |
93.16 |
92.84 |
0.48 |
0.77 |
|
2.24 |
2.06 |
91.79 |
||||
Resorcinol |
0.52 |
0.47 |
93.65 |
|||
0.65 |
0.61 |
91.54 |
93.03 |
1.01 |
0.88 |
|
0.78 |
0.74 |
94.97 |
Table 3 Results of the determination of the sample
Components |
Marked content /% |
Average determined content /% |
Salicylic acid |
- |
4.54 |
3. CONCLUSION
REFERENCES
[1] Guo Xingjie, Zhou Mi. Chinese Journal of Chromatography (Se Pu), 1998, 16 (6) :
532-533.
[2] Yang Yaofang, Qi Jingli,He Beili et al. China
Hospital Pharmaceutical Journal (ZhongguoYiyuan Yaoxue Zazhi) ,2000,20 (9): 568-569.
[3] Pirker R, Huck CW, Popp M et al. Journal of Chromatography B, Analytical Technologies
in the Biomedical and Life Sciences, 2004, 809 (2): 257-264.
[4] Xing Rong,Deng Ziyu,Chen Gende. China Hospital Pharmaceutical Journal (ZhongguoYiyuan
Yaoxue Zazhi),2005, 25 (3): 227-229.
[5] Franeta J T, Agbaba D, Eric S et al. IL Farmaco, 2002, 57 : 709-713.
[6] Gomez, Maria R, Olsina et al. Talanta, 2003, 61 (2): 233-238.
[7] Engelberth J, Schmelz E A, Alborn H T et al. Anal.Biochem., 2003, 312: 242.
张前莉 周建科 韩康 张立
(河北大学理化分析中心,河北省分析科学技术重点实验室,保定071002)
摘要 建立了复方醋酸氟轻松酊中水杨酸、苯酚、间苯二酚的反相离子对色谱测定法。采用反相C18柱(150*4.6mm i.d., 5mm),甲醇-乙腈-水10:25:65(v/v/v)、四丁基溴化铵浓度为1.0mmol/l的混合溶液做流动相, 225nm波长下检测。水杨酸、苯酚、间苯二酚平均回收率均达到85.0%以上;在0.014mg/ml-5.40μg/ml范围内,线性相关系数分别为0.9971,0.9997,0.9977;检出限分别为: 0.019μg/ml, 0.027 mg/ml, 0.014mg/ml;相对标准偏差在0.48%-3.34%之间。
关键词 复方醋酸氟轻松酊;水杨酸;苯酚;间苯二酚;反相离子对色谱