http://www.chemistrymag.org/cji/2006/087048pe.htm |
Jul. 1, 2006 Vol.8 No.7 P.48 Copyright |
Du Xinzhen1,2, Tao Xiaojuan, Wang
Yarong1, Ding Ning1, Chen Hui1
(1 Department of Chemistry, Northwest Normal University, Lanzhou 730070; 2
The Key Laboratory of Polymer Materials of Gansu Province, Lanzhou 730070, China)
Received Mar.10, 2006;Supported by the National Natural Science Foundation of China (No: 20575053), the Natural Science Foundation of Gansu Province (3ZS051-A25-011-022) and the Key Laboratory of Polymer Materials of Gansu Province.
Abstract Mesoporous silica and
composite were used as fiber coatings to evaluate their adsorptive properties. A
diffusion-controlled process was involved for the adsorption of toluene, p-xylene,
biphenyl and anthracene onto mesoporous materials in water matrix. Vigorous stirring and
heating of the solution are favorable for the diffusion of the studied compounds. The
fiber coated with phenyl-MCM-41 shows high adsorption efficiency and good molecular
recognition but negative effect of salt on adsorption of target compounds. Rapid
desorption could be obtained in aqueous solution containing methanol. The fiber is easy to
prepare and handle. Moreover, only a small quantity of adsorption material was needed to
prepare the sorption fibers compared with conventional batch sorption experiments. The
fibers are inexpensive, durable and easy to couple with HPLC. Quantitative adsorption and
desorption was obtained with good reproducibility. The coated fibers offer an alternative
way to evaluate the adsorption behavior of potential adsorption or separation materials.
Keywords Adsorption; mesoporous silica; sorption fibers; aromatic compounds
1. INTRODUCTION
Since the discovery of the novel M41S mesoporous materials [1], extensive
attention was paid to them to be served as adsorbents and separation materials because
they have larger specific surface area and uniform mesostructure [2]. Organic
compounds and metal ions can selectively adsorb onto the surface of mesoporous materials [3-7],
especially mesoporous composites [8-12]. For the purpose of better
understanding of adsorption behavior of synthesized materials, batch sorption experiments
of target compounds onto the adsorbents were usually performed by adding a certain amount
of adsorbent (20~200mg) to a solution which is stirred continuously or allowed to stand
for 10-24 h on a platform shaker to ensure that the adsorption equilibrium is reached. The
slurries were centrifuged or filtered. Subsequently the concentration of target compounds
in the supernatants or filtrates were measured using conventional analytical techniques.
The amount of specifically adsorbed compounds was calculated by the difference between
initial and final concentration of corresponding compound in solution. The procedures are
tedious and expensive. For these reasons, an attempt was made to use MCM-41 mesoporous
silica as the coatings of sorption fibers which integrate the adsorption, concentration
and sample introduction in a single step. The cylindrical surface geometry of the fibers
is well defined and allows easy access of target compounds to and from the surface,
leading to efficient adsorption and desorption. The miniature dimensions of the fiber are
also convenient to couple with HPLC [13, 14]. Several factors affecting the
adsorption and desorption properties were studied. The adsorption efficiency and
selectivity were also discussed.
2. EXPERIMENTAL
2.1. Materials
Tetraethyl orthosilicate (Shanghai Chemical Reagents Co.), cetyltrimethylammonium
bromide (Beijing Chemical Reagents Co.), toluene (To) and p-xylene (Xy) (Tianjin
Chemical Reagents Co.), biphenyl (Bp) and anthracene (An) (China National Medicines Co.),
sodium chloride (Shanghai Hunter Fine Chemicals Ltd.) and trimethoxyphenylsilane (Fluka)
were of analytical grade. Pure mesoporous silica was synthesized following the procedures
of the literature [15]. The removal of template was carried out in ethanol
containing 1 mol·L-1 hydrochloric acid.
One-step synthesis of phenyl functionalized MCM-41 was improved by consulting with the
literature [16]. Doubly distilled water was used.
2.2. Analysis and methods
The structure of the synthesized material was characterized by a D8 diffratometer
(Bruker, Germany). Identification of organic functional groups was performed on 670
infrared spectrometer (Nicolet, USA) and the percentage of C and H was estimated by 2400
CHN elemental analyzer (Perkin-Elmer, USA). The specific surface area was determined by
the Brunauer-Emmett-Teller on an ASAP 2010 instrument (Micromeritics, USA). The measured
physical parameters of the synthesized materials were listed in Table 1. The thermal
stability of mesoporous composite was examined on D4 thermogravimetric system (Shimazdu,
Japan). The thickness of fiber coatings was measured using micrometer caliper. The coated
fiber was characterized by a JSM-5600LV scanning electron microscope (JEOL, Japan). The
efficiency of adsorption was estimated on LC-6A liquid chromatograph (Shimazdu, Japan)
with a Waters C18 column and SPD-6AV UV-Vis absorbance detector.
Table 1 Physical parameters of synthesized mesoporous materials
Materials |
Surface area (m2·g-1) |
Carbon load (%) |
Pore size (nm) |
Phenyl-MCM-41 |
927.89 |
30.10 |
2.13 |
2.3. Preparation of sorption fibers
The fiber was modified from a commercial 1-ml HPLC syringe. The plunger wire (160 mm o.d.) inside the needle was cleaned
with ethanol and dried at 60oC. The mesoporous particles were immobilized onto
the steel wire with epoxy glue. The coated plunger wire was heated for cure at 80oC
for 8 hours. The fiber coating is 10 mm long and 10 mm thick. Fig. 1 shows SEM image of the
sorption fiber coated with phenyl-MCM-41. The coated fibers were conditioned in mobile
phase of HPLC for 30 min to free from contaminants prior to use.

Fig. 1 Scanning electron micrograph of phenyl-MCM-41
coated fiber.
2.4. Procedures
The protecting needle of the fiber was pierced into the glass bottle sealed with a
silicone septum and the coated fiber was exposed to the stirred solution containing To,
Xy, Bp and An of 1.00×10-6 mol× L-1 for adsorption respectively.
The adsorption was carried out on a heated stirring platform with the magnetic stirring
rate of 1000 rpm at 20 oC. The needle holding the coated fiber was withdrawn
from the water matrix and introduced into desorption chamber connected with six-port
injection valve of HPLC for 5-min static desorption in methanol/water (70/30 v/v) when the
injection valve was in the load position. Subsequently the valve was switched to the
injection position for the delivery of target compounds to the chromatographic column at
the flow-rate of 1 ml× min-1. Chromatographic peak area was utilized to
examine direct adsorption efficiency of mesoporous materials.
3. RESULTS AND DISCUSSION
3.1 Adsorption
Fig. 2 shows time dependence of the adsorption process for the fibers with MCM-41 and
phenyl-MCM-41. A period of time was practically needed to reach their adsorption
equilibrium for all of the compounds. Adsorption equilibrium of MCM-41 coating was almost
established within 60 min. When phenyl-MCM-41 was used as the fiber coating, the amount
adsorbed was increased but longer adsorption time was required to reach equilibrium. The
equilibrium time for adsorption process increases with decreasing pore size of mesoporous
materials and with increasing molecular size of target compounds. Actually it is not
necessary to reach the equilibrium for adsorption process. The adsorption time of 30 min
is a reasonable compromise between chromatographic peak area and adsorption time for
MCM-41 and phenyl-MCM-41 coatings.
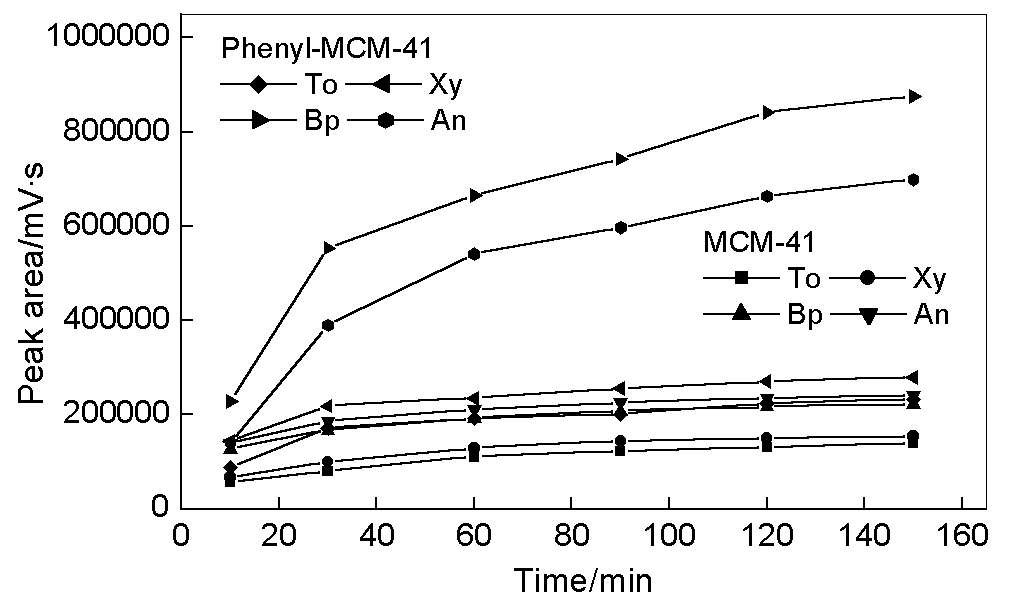
Fig. 2 Dependence of the adsorption on time.
3.2 Desorption
Solvent desorption of the adsorbed compounds in the injector is the reverse process of
adsorption. Fig. 3 gives typical desorption time profiles of phenyl-MCM-41 coating in
methanol/water. There is a little mass transfer resistance during the desorption process
which is much fast compared to that of adsorption process. 1-min and 3-min are enough to
reach the equilibrium of desorption for To and Xy as well as Bp and An, respectively. The
ratio of methanol and water in mobile phase has a significant effect on the desorption
process. Large amount content of methanol in mobile phase leads to more rapid desorption.
This is supportive of the fact that adsorption and desorption processes are dynamically
controlled by diffusion in the mesoporous materials.
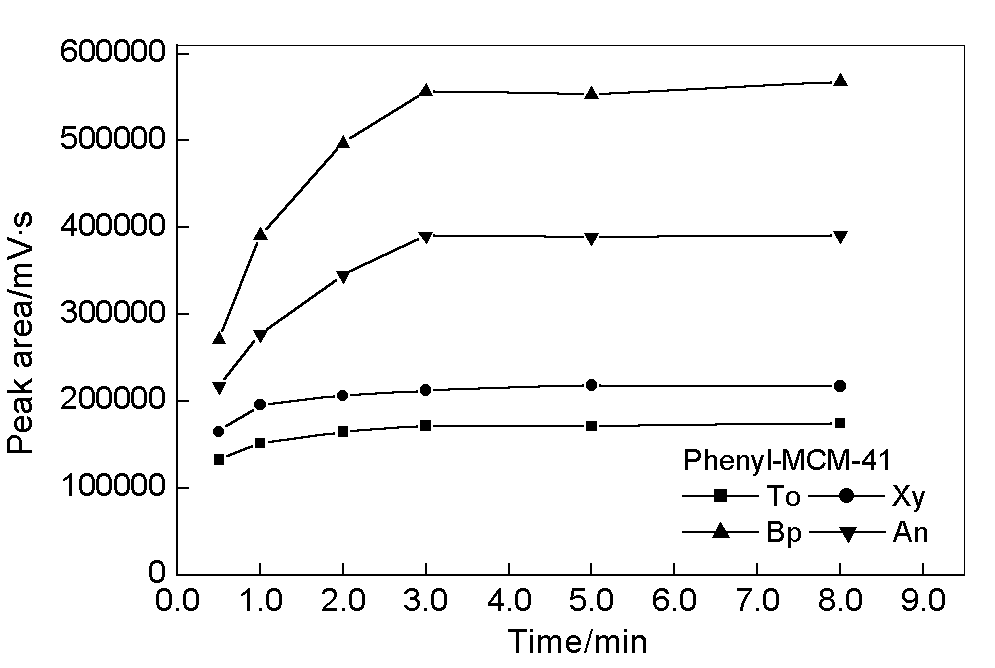
Fig. 3 Dependence of the desorption on time.
Adsorption time, 30 min.
3.3 Mass transfer
Stirring is very important because adsorption is a dynamic diffusion-controlled process.
Fig. 4 compares the dependence of adsorption on stirring rate. The adsorption of MCM-41
coating is faster in the stirred solutions and 1000 rpm is enough to approach perfect
stirring. For the phenyl-MCM-41 coating, however, the adsorption process greatly depends
on the degree of agitation. As a result of small pore size of phenyl-MCM-41, one can infer
that the diffusion process is slower because perfect stirring is difficult to be achieved
in the smaller mesopores of phenyl-MCM-41. There is larger resistance of mass transfer for
the diffusion of the adsorbed compounds from bulk solution into the smaller pores of
phenyl-MCM-41 than that of MCM-41, especially for the diffusion of Bp and An.
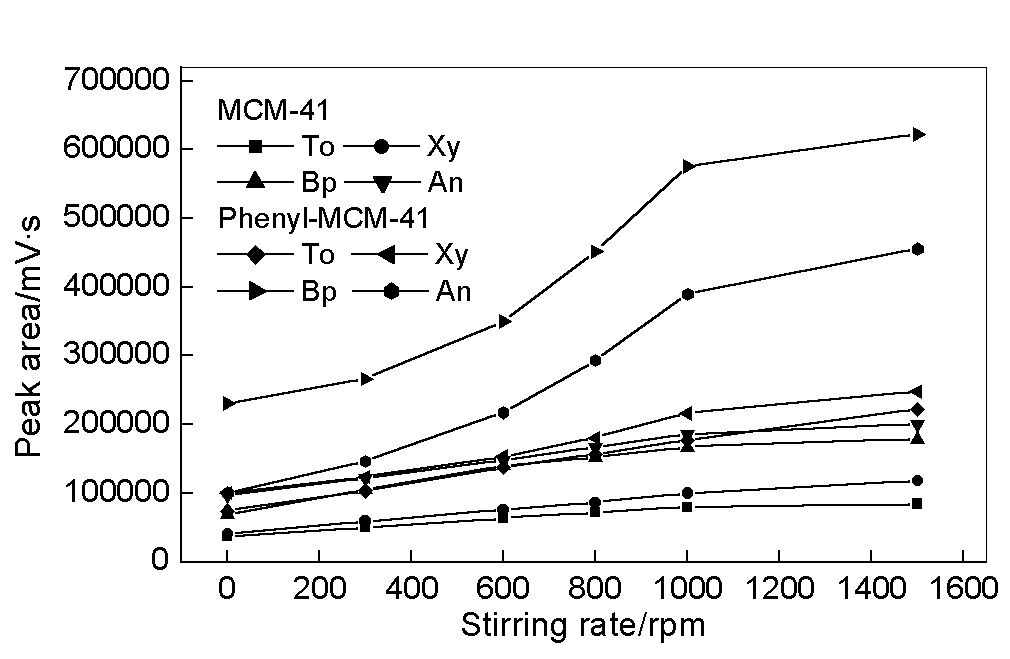
Fig. 4 Dependence of the adsorption on stirring rate. Adsorption time, 30 min.
Generally temperature also plays an important role in adsorption process because of its potential influence on thermodynamics and kinetics of adsorption process of target compounds between fiber coating and water matrix. High temperature is unfavorable to adsorption of target compounds because adsorption is generally an exothermic process. However, both MCM-41 and phenyl-MCM-41 coated fibers presented a positive effect of temperature on adsorption of target compounds. Furthermore the adsorption efficiencies increased with the increasing temperature of water matrix to a greater extent for phenyl-MCM-41 coating. It clearly indicates that mass transfer is the predominant factor during adsorption process for mesoporous materials. For the sake of avoiding the volatility of aromatic compounds, 20 ℃ was employed.
3.4 Ionic strength
Fig. 5 shows the dependence of adsorption on the ionic strength of water matrix for phenyl-MCM-41 coated fiber. The amount adsorbed decreases with increasing concentration of the salt. This result may arise from the increased ionization of silanols at the surface of mesoporous coating by the addition of sodium ions [17]. The concentration of adsorbed sodium ions at the liquid-solid interface is higher than that in bulk solution, which changes the physical properties in the mesopores. Consequently higher concentration of sodium chloride results in lower concentration of target compounds at the interfacial area compared to the bulk solution because higher concentration of salt causes a decreased solubility of nonpolar compounds in water matrix. On the other hand, addition of salt can increase the viscosity of aqueous solution, especially the solution at liquid-solid interface. This may limit the diffusion of target compounds from bulk solution to the mesoporous surface of fiber coating and result in lower adsorption efficiency of studied compounds.
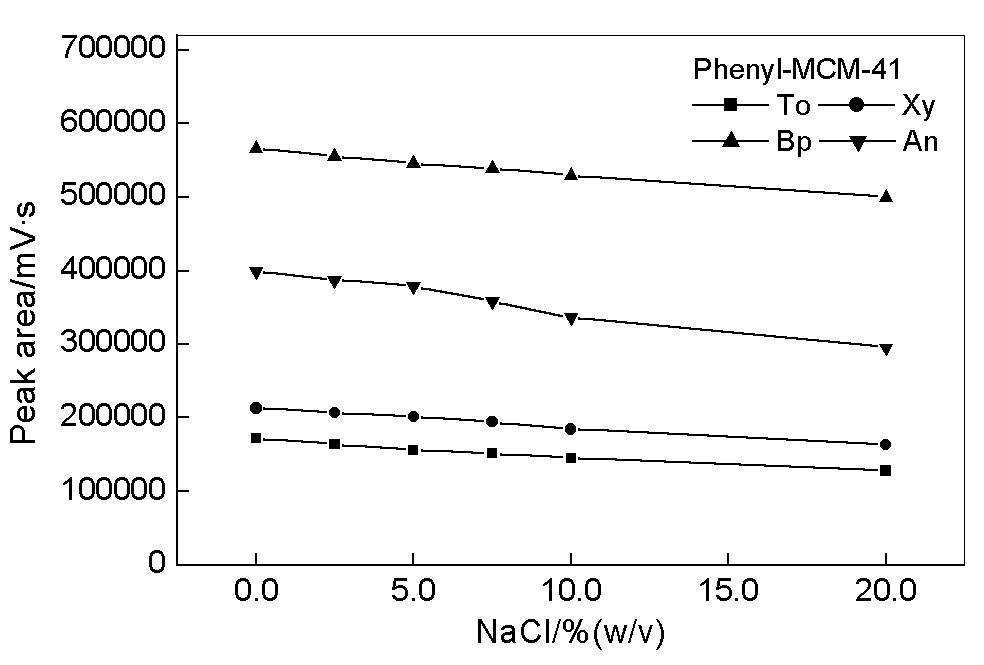
Fig. 5 Dependence of the adsorption on ionic strength. Adsorption time, 30 min.
3.5 Efficiency and selectivity of adsorption
The adsorption efficiency of mesoporous materials depends on the partitioning of the
target compound between fiber coating and water matrix. As shown in Fig. 2, high
concentration of the studied compounds was obtained on the phenyl-MCM-41 fiber than the
MCM-41 one. Table 2 summarizes the distribution constants (KD) of four
compounds. Adsorption efficiency of MCM-41 coating suggests that larger surface area plays
a sole role in adsorption process. The polar surface of MCM-41 shows decreasing affinity
in the order: To>Xy>Bp>An, in agreement with that of their solubility in water.
After chemical modification at the surface of MCM-41, adsorption efficiency of
phenyl-MCM-41 coating was about 2~4 times greater than that of MCM-41 coating although the
surface area of phenyl-MCM-41 decreases to some extent compared to that of MCM-41.
Clearly, surface modification also greatly contributes to higher adsorption efficiency.
However, it should be noted that the adsorption behavior of
phenyl-MCM-41 coating was very different from that of MCM-41 coating. As shown in Table 2,
the KD values for toluene and biphenyl are larger than those for
p-xylene and Anthracene on the phenyl-MCM-41 coating, respectively. This indicates that
selectivity of adsorption was achieved by hydrophobic nature and smaller mesopores in
adsorption process when the mesoporous surface was chemically bonded with phenyl group.
Surface modification is thermodynamically favorable to the adsorption process of Bp and An
with low solubility but results in larger resistance of mass transfer from bulk solution
to smaller mesopores. Thereby it takes longer time to reach adsorption equilibrium for Bp
and An.
Table 2 Distribution constants of aromatic compounds
Phase type |
KD |
|||
Toluene |
p-Xylene |
Biphenyl |
Anthracene |
|
Phenyl-MCM-41 |
1071 |
873 |
1692 |
902 |
3.6 Stability of sorption fibers
Ryco et al reported that the structure of MCM-41 mesoporous silica was completely lost
upon boiling in water for two days due to silicate hydrolysis [18]. In batch
sorption experiments, the structural collapse may have a significant effect on the
adsorption behavior of materials. According to the procedures described, the cylindrical
surface geometry of the fibers allows easy access of target compounds to and from the
surface, leading to efficient adsorption and desorption in a short time. Furthermore,
phenyl-silylation of MCM-41 effectively enhances the hydrothermal stability. The custom
made fiber with phenyl-MCM-41 coating can at least withstand 200 adsorption-desorption
cycles toward vigorous stirring and desorption under the conditions employed. The lifetime
of the phenyl-MCM-41 coating becomes much longer than that of pure mesoporous silica
coating. Relative standard deviation of five replicate adsorption-desorption runs is
0.10%~1.60% for To, Xy, Bp and An of 1.00×10-6 mol× L-1 in spiked
water.
4. CONCLUSIONS
The fibers coated with MCM-41 and phenyl-MCM-41 were prepared to examine the
adsorptive properties of the synthesized materials. The adsorption of mesoporous coatings
involves a diffusion-limited process. The phenyl-MCM-41 coating shows greater adsorption
efficiency and better selectivity of target compounds than MCM-41 one. A regeneration of
the fiber coating can be performed by desorption in methanol/water and the original
adsorption efficiency was obtained again. The sorption fibers can tolerate many
adsorption-desorption cycles under the experimental conditions. As compared with
conventional batch sorption experiments, the sorption fiber was prepared with the
adsorption material of less than 10 mg. Rapid and quantitative adsorption and desorption
of target compounds can easily be achieved with good reproducibility by coupling HPLC.
Consequently the sorption fiber is a useful tool for obtaining information about potential
adsorption or separation materials.
REFERENCES
[1] Kresge C T, Leonowicz M E, Roth W J. et al. Nature, 1992, 359: 710.
[2] Beck J S, Vartuli J C, Roth W J et al. J. Am. Chem. Soc., 1992, 114: 10834.
[3] Feng X, Fryxell G E, Wang L Q et al. Science, 1997, 276: 923.
[4] Liu A, Hidauzt K, Kawi S et al. Chem. Commun., 2000, 1145.
[5] Vartuli J C, Malek A, Roth W J et al. Micropor. Mesopor. Mater., 2001, 44-45: 691.
[6] Nassivera T, Eklund A G, Landry C C. J. Chromatogr. A, 2002, 973: 97.
[7] Shindo T, Kudo H, Kitabayashi S et al. Micropor. Mesopor. Mater., 2003, 63: 97.
[8] Dai S, Burleigh M C, Shin Y et al. Angew. Chem. Int. Ed., 1999, 38: 1235.
[9] Lee H, Yi J. Sep. Sci. Technol., 2001, 36: 2433.
[10] Hou J G, Ma Q, Du X Z et al. Talanta, 2004, 62: 241.
[11] Ghiaci M, Abbaspur A, Kia R et al. Sep. Purif. Technol., 2004, 40: 217.
[12] Im H J, Barnes C E, Dai S et al. Micropor. Mesopor. Mater., 2004, 70: 57.
[13] Chen J, Pawliszyn J B. Anal. Chem., 1995, 67: 2530.
[14] Liu Y, Lee M L, Hageman K J et al. Anal. Chem., 1997, 69: 5001.
[15] Buchel G, Denoyel R, Llewellyn P L et al. J. Mater. Chem., 2001, 11: 589.
[16] Fowler C E, Burkett S L, Mann S. Chem. Commun., 1997, 1769.
[17] Khurana A L, Ho C T. J. Liquid Chromatogr., 1998, 11: 3205.
[18] Ryco R, Kim J M, Ko C H et al. J. Phys. Chem., 1996, 100: 17718.
杜新贞, 陶小娟, 王亚荣, 丁宁, 陈慧
(西北师范大学化学系, 兰州 730070)
摘要 以介孔二氧化硅及其复合体作为探头涂层评价了介孔材料的吸附性质,在水基体中,介孔材料对甲苯、对二甲苯、联苯和蒽的吸附过程受扩散控制,充分搅拌和加热有利于所研究芳烃分子的扩散。phenyl-MCM-41探头涂层有高的吸附效率和良好的分子识别能力,但盐效应不利于吸附,探头吸附物在甲醇/水溶液中可快速解吸。该探头易于制作,而且与常规批量吸附试验相比所需吸附材料很少,成本低,可多次重复作用,可与HPLC联用,定量吸附和解吸重现性好,为潜在吸附分离材料的评价提供了一种选择途径。
关键词 吸附; 分离; 介孔二氧化硅; 吸附探头; 芳香烃