http://www.chemistrymag.org/cji/2006/087049pe.htm |
Jul. 1, 2006 Vol.8 No.7 P.49 Copyright |
Yu Qing, Bao Xiaoge, Zhu Ligang, Bian Hedong,
Liang Hong
(College of Chemistry and Chemical Engineering, Guangxi Normal University, Guilin 541004,
China)
Keywords Schiff base, zinc(II) complex, pyridine-2-carbaldehyde semicarbazone, crystal structure Thiosemicarbazones and their metal complexes are of considerable interest because of their potential pharmacological properties[1], and a wide variation in their modes of bonding and stereochemistry[2] whereas their semicarbazone analogs have been received much less attention[3]. However, semicarbazones are reported to possess versatile structural features and very good antifungal and antibacterial properties. Three structural isomers (E-, E'- and Z-forms) are reported[4], and they can coordinate to the metal either as a neutral ligand or as a deprotonated ligand through the NNO atoms[4-5]. In our continuing studies[6-7], the title complex of the Schiff base (PASC) that are synthesized from the condensation of pyridine-2-carbaldehyde and semicarbazone had been reported[8].
1. EXPERIMENTAL
1.1 Materials and physical measurements
All reagents were of analytical grade
from commercial sources and were used without any further purification. The PASC was
prepared according to the reference[8]. Elemental analyses for C, H and N atoms were carried out
on a Model 2400 IIPerkin-Elmer elemental analyzer.
The infrared spectrum was taken on a Perkin-Elmer Spectrum One FT-IR Spectrometer in the
4000--400 cm-1 region by using KBr pellets. The diffraction data were collected
on a Bruker SMART CCD diffractometer using graphite monochromated MoK
1.2 Preparation of [Zn(PASC)2](ClO4)2·H2O
Single crystals of the title complex suitable for X-ray crystallographic analysis were obtained by solvothermal treatment of Zn(ClO4)2·6H2O (0.0744 g, 0.2 mmol) and the Schiff base ligand (0.0328 g, 0.2 mmol), methanol (0.2 mL) and ethanol (1.0 mL). The mixture was placed in a thick Pyrex tube (ca 20 cm long), then cooled with liquid N2, and air evacuated. The sealed tube was heated at 353K for two days to yield colorless crystals. Anal.calcd.(found) for C14H18Cl2N8O11Zn: C, 27.54 (27.48); H, 2.95 (2.98); N, 18.36 (18.28)%. IR (KBr, cm-1): 3436(s), 1662(ms), 1633(ms), 1474(m), 1108(s), 774(w), 627(m).
1.3 Crystal structure determination and refinement
Suitable crystals of the title complex (colorless, dimensions 0.26 mm × 0.24 mm × 0.20 mm) was used for the structure determination. Data collection at 293(2) K was carried out with a Bruker SMART CCD instrument using MoKa radiation. The structures were solved by direct methods in SHELXS-97[9] and refined by full-matrix least-squares, based on F2, using SHELXL-97[10]. All non-hydrogen atoms were assigned anisotropic thermal parameters. The coordinates of the hydrogen atoms were obtained from difference Fourier maps and refined with a common isotropic thermal parameter. For the title complex, convergence was reached at a final R1 = 0.0387 [for I > 2s(I)], wR2 = 0.1115 (for all data), with allowance for the thermal anisotropy for all non-hydrogen atoms. Details of data collection and processing are given in Table 1.
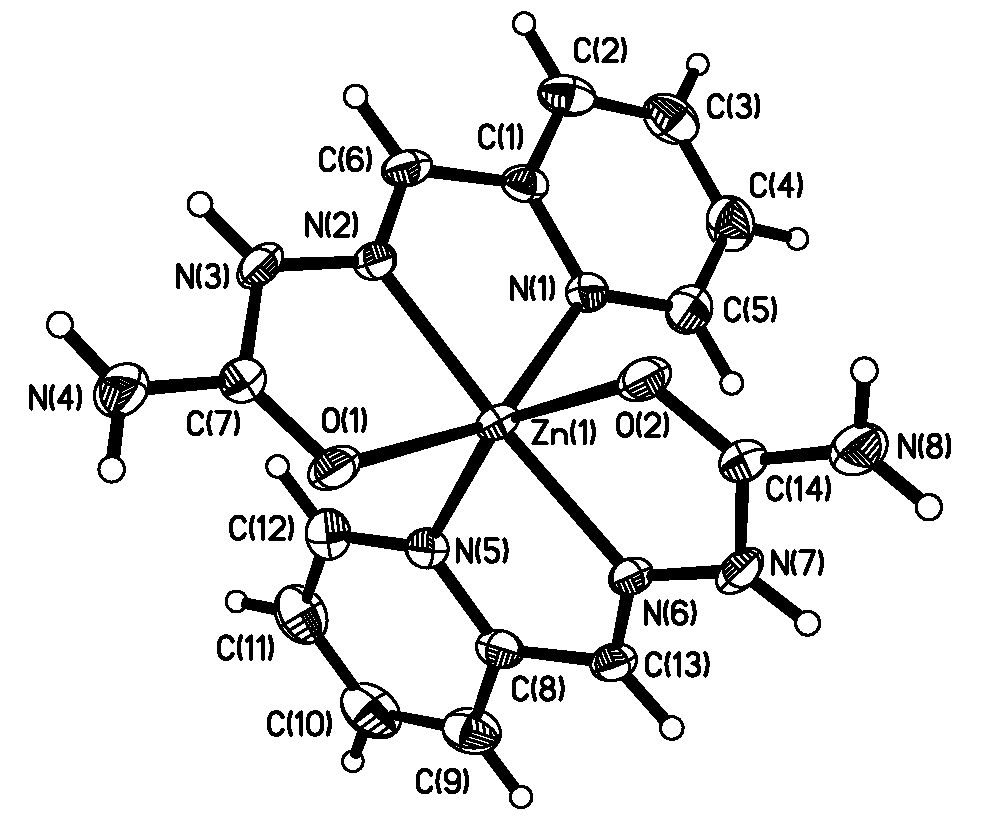
Fig.1 ORTEP projection of [Zn(PASC)2]2+
Table 1 Crystal data and structure analysis parameter of the complex
| Complex | [Zn(PASC)2](ClO4)2·H2O |
V (nm3) |
1.1653(3) |
| Crystal size (mm3) | 0.26 × 0.24 × 0.20 | Z | 2 |
| Empirical formula | C14H18Cl2N8O11Zn | Dc (g/cm3) | 1.740 |
| Temperature (K) | 293(2) | F (000) | 620 |
| Wavelength (nm) | 0.071073 | Scan mode | w-2q |
| Formula weight | 610.63 | range for data collection (°) | 2.02-26.42 |
| Crystal system | triclinic | Absorption coefficient(mm-1) | 1.356 |
| Space group | Pī | Reflections collected | 6734 |
| a (nm) | 1.0064(2) | Independent reflections | 4677 |
| b (nm) | 1.1236(2) | Goodness-of-fit | 1.044 |
| c (nm) | 1.2252(2) | R [I > 2s (I)] | 0.0387 |
| a(°) | 113.017(6) | wR2 | 0.1115 |
| b(°) | 110.058(7) | Maximum diff. peak (e/nm3) | 489 |
| g(°) | 93.780(6) | Minimum diff. peak (e/nm3) | -361 |
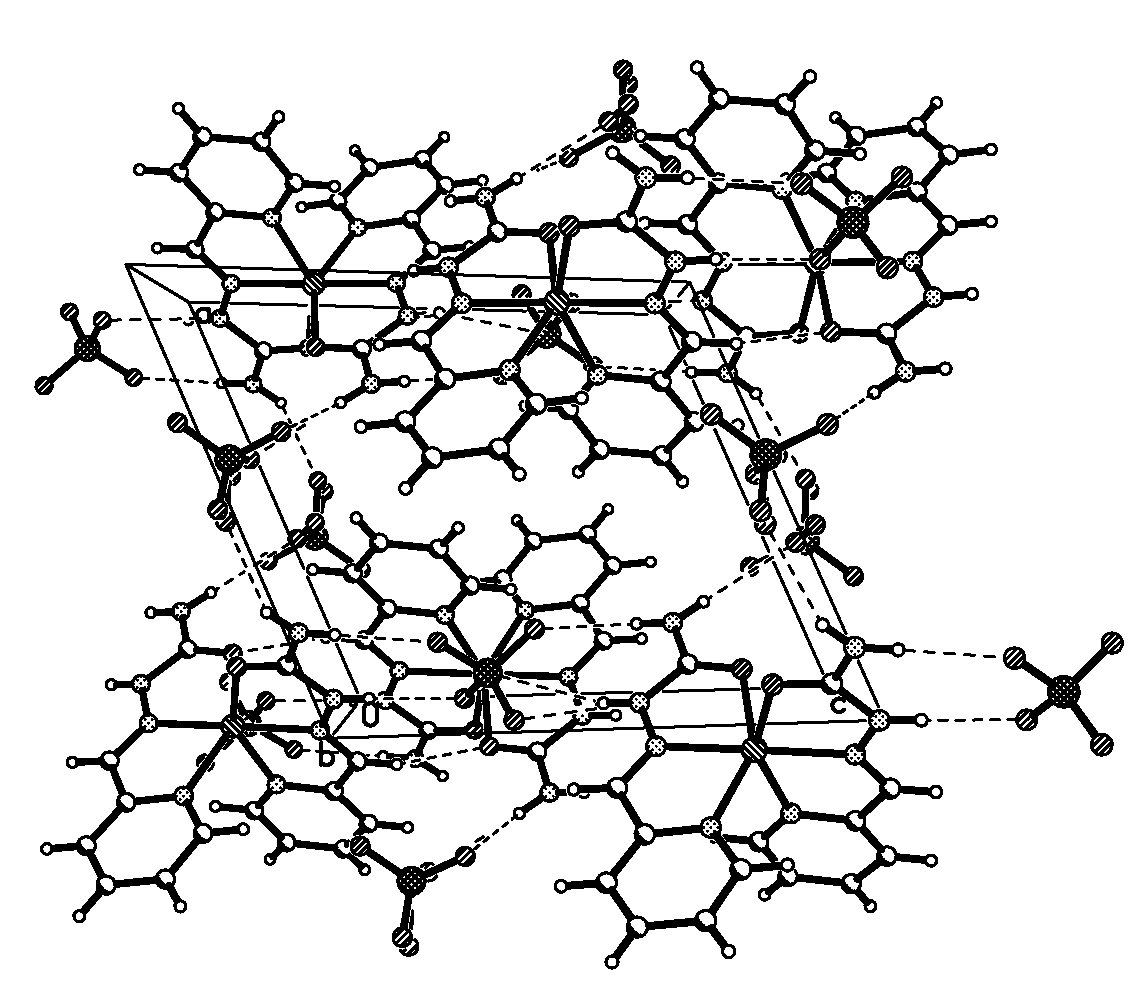
Fig.2 The 2-D network structure by hydrogen bonds of the title complex Table 2 Selected bond lengths (nm) and bond angles (°) of the complex
| Bonds | Aengths |
Bonds |
Lengths |
| Zn(1)-N(1) | 0.2189(2) | Zn(1)-N(6) | 0.2079(2) |
| Zn(1)-N(2) | 0.2070(2) | Zn(1)-O(1) | 0.2160(2) |
| Zn(1)-N(5) | 0.2151(3) | Zn(1)-O(2) |
0.2176(2) |
| Bonds | Angles |
Bonds |
Angles |
| N(2)-Zn(1)-N(6) | 176.17(9) | N(5)-Zn(1)-O(2) | 149.22(9) |
| N(2)-Zn(1)-N(5) | 106.09(9) | O(1)-Zn(1)-O(2) | 92.48(9) |
| N(6)-Zn(1)-N(5) | 75.85(9) | N(2)-Zn(1)-N(1) | 75.54(9) |
| N(2)-Zn(1)-O(1) | 74.32(9) | N(6)-Zn(1)-N(1) | 101.06(9) |
| N(6)-Zn(1)-O(1) | 109.01(9) | N(5)-Zn(1)-N(1) | 96.04(9) |
| N(5)-Zn(1)-O(1) | 93.73(9) | O(1)-Zn(1)-N(1) | 149.82(9) |
| N(2)-Zn(1)-O(2) | 104.61(8) | O(2)-Zn(1)-N(1) | 93.55(9) |
| N(6)-Zn(1)-O(2) | 73.62(9) |
2. RESULTS AND DISCUSSION
The structure of the title complex consists of discrete [Zn(PASC)2]2+
cations, perchlorate counter-ions and water molecules.Figure 1 shows the ORTEP projection
of [Zn(PASC)2]2+,and selected bond lengths and bond angles are
listed in Table 2. The two neutral semicarbazone ligands are planar, and each set of the
coordinating carbonyl O, imino N and pyridyl N atoms occupies a meridional plane of the
octahedron around the Zn atom. The Zn1/O1/C7/N3/N2/C6/C1/N1 (mean deviation from plane =
0.04795 nm) and Zn1/O2/C14/N7/N6/C13/C8/N5 (mean deviation from plane = 0.00239 nm) planes
are nearly perpendicular to each other (dihedral angle 81.2o). The
perchlorate counter-ion is not involved in coordination, a feature that is also observed
in bis{1-[(E)-2-pyridinylmethylidene] semicarbazide} cobalt(II) diperchlorate
monohydrate[11]. .One perchlorate anion is much disordered, with the oxygen
atoms occupying eight sites. The pyridine-2-carbaldehyde semicarbazone ligands possess an E
configuration with respect to the azomethine double bond, which is different from
bis(2-formylpyridine semicabazonato)nickel(II) [11].
The Zn-N and Zn-O coordination distance are comparable
with those reported for other zinc(II) complexes with 2-acetylpyridinesemicarbazone or
2,6-diacetylpyridine bis(semicarbazone) [2]. The
C=O bond lengths [C(7) - O(1) = 0.1234(4) nm and C(14) - O(2) = 0.1240(3) nm] are close to
those reported by Liang et al [6]. The distortions from ideal octahedral
coordination of the zinc(II) are seen in the chelate bite angles, which are less than 90o.
The supramolecular architecture is stabilized by an extensive 2-D network (Fig.2, Table 3)
of hydrogen bonds (N-H···O) involving the cations, counter-ions and
water molecules.
Table 3 Hydrogen-bonding geometry (nm, °) for the
title complex
| D-H···A | ARU |
d(D-H)/nm |
d(H ···A)/nm |
d(D ···A)/nm |
∠(DHA)/° |
N(3)-H(3A) ···O(8) |
2555.03 |
0.086 |
0.209 |
0.29033 |
158 |
N(4)-H(4A) ···O(6) |
1445.02 |
0.086 |
0.254 |
0.31438 |
128 |
N(4)-H(4A) ···O(5) |
2555.02 |
0.086 |
0.254 |
0.32236 |
137 |
N(4)-H(4B) ···O(7) |
2555.03 |
0.086 |
0.222 |
0.30672 |
168 |
N(7)-H(7A) ···O(9) |
2566.03 |
0.086 |
0.199 |
0.28256 |
164 |
N(8)-H(8A) ···O(5) |
2565.02 |
0.086 |
0.257 |
0.33081 |
144 |
N(8)-H(8B) ···O(10) |
2566.03 |
0.086 |
0.224 |
0.30600 |
159 |
ARU-code: 2555. = -x, -y, -z; 1445. = -1+x, -1+y, z; 2566. = -x, 1-y, 1-z; 2565. = -x, 1-y, -z.
REFERENCE
[1] West D X, Liberta A E, Padhye S B et al. Coord. Chem. Rev, 1993, 123: 49-71.
[2] Kasuga N C, Sekino K, Ishikawa M et al. J. Inorg. Biochem, 2003, 96: 298-310.
[3] Sreekanth A, Kala U L, Nayar C R et al. Polyhedron, 2004, 23: 41-47.
[4] West D X, Bain G A, Butcher R J et al. Polyhedron, 1996, 15: 665-674.
[5] Casas J S, Garcia M S, Sordo J. Coord. Chem. Rev, 2000, 209: 197-261.
[6] Liang H, Chen X B, Hu R X et al. Acta. Chim. Sinica, 2002, 60: 939-946.
[7] Zhou J, Chen Z F, Tan Y S et al. Acta. Cryst. E, 2004,60: 519-521.
[8] Gong Y Q, Chen C G, Huan X F. Synth. React. Inorg. Met. Org. Chem, 1994,24: 877-889.
[9] Sheldrick G. M. SHELXS-97, Program for X–ray
Crystal Structure Solution. University of Göttingen: Germany, 1997.
[10] Sheldrick, G. M. SHELXL-97, Program for X–ray
Crystal Structure Refinement. University of Göttingen: Germany, 1997.
[11] Zhou J, Chen Z F, Wang X W et al. Acta. Cryst. E, 2004,60: 568-570.
于青 鲍晓鸽 朱立刚 边贺东 梁宏
(广西师范大学化学化工学院,桂林 541004,中国)
摘要 本文以吡啶-2-甲醛缩氨基脲(PASC)为配体合成了锌(II)配合物[Zn(PASC)2](ClO4)2·H2O,并测定了其晶体结构。该单晶属三斜晶系,空间群为Pī,晶胞参数a = 1.0064(2) nm, b = 1.1236(2) nm, c = 1.2252(2) nm, a = 113.017(6)o, b = 110.058(7)o, g = 93.780(6)o, V = 1.1653(3) nm3, wR2 = 0.1115, Z = 2。衍射数据(I > 2s (I))为4677。晶体结构表明,配合物中Zn2+处于一个畸变的八面体配位环境,配位原子分别来自配体中吡啶环上的氮原子,亚胺基氮原子以及羰基氧原子。
关键词 席夫碱, 锌(II)配合物, 吡啶-2-甲醛缩氨基脲, 晶体结构